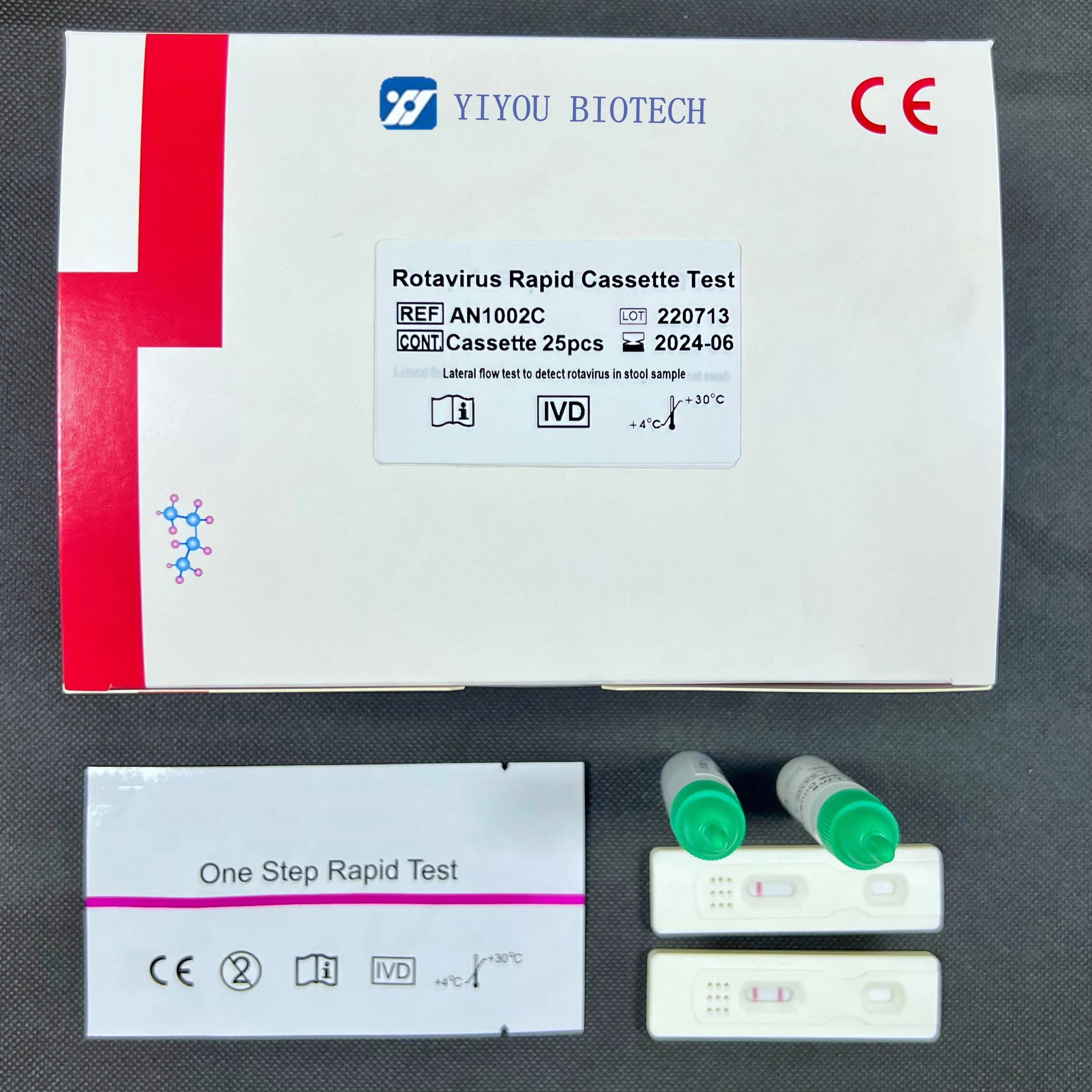
Diagnostic Test Kits Rotavirus Rapid Test for qualitative Detection of Rotavirus Antigen in Feces
- Category: Custom Chemical Services >>>
- Supplier: Biocare Diagnostics Ltd.
Share on (11000005836588):
Product Overview
Description
Specification
Brand Name | Biocare |
Product name | Rotavirus Rapid Test |
Package Size | 25tests/kit, 10tests/kit |
Specimen | human feces |
Brand Name | Biocare or OEM |
Shelf Life | 24months |
Certificate | ISO13485 |
Storage | 4-30℃ |
Product Description
Rotavirus Rapid Test Card
Immunochromatographic rapid test card for qualitative detection of rotavirus antigen in feces
INTRODUCTION
Rotaviruses are the main cause of acute gastroenteritis, especially in children under the age of two years. Rotaviruses have been identified in almost 50% of the faeces of children with gastroenteritis, responsible for 600,000-800,000 death annually. Rotavirus infections occur frequently during the winter months. Gastroenteritis from enteric viruses can be mortal in risk populations such as children, the elderly or immunosuppressed individuals. Characteristic symptoms include vomiting, hydrodiarrhoea for between 3 and 8 days, high temperature and stomach pains. Rotaviruses transferred via the faecal-oral route are eliminated in large quantities into the intestine, so that hospital-borne infections from rotaviruses are regarded very seriously, particularly in baby stations and paediatric clinics, and are difficult to control. Early and reliable detection so that rotaviruses can be recognised and further infections avoided is therefore very important.
PRINCIPLE OF THE TEST
The Rotavirus Rapid Test Device employs red gold-conjugated monoclonal antibodies against antigen VP6 of group A of rotavirus, and solid-phase specific rotavirus antibodies. In this test the specimen is first treated with an extraction solution to extract rotavirus antigens from the faces. Following extraction, the only step required is to add the extract to the reaction device. As the sample flows through the test membrane, the colored particles migrate. In the case of a positive result the specific antibodies present on the membrane will capture the colored particles. Red color lines will be visible, depending upon the virus content of the sample. These lines, after 5-15 minutes of incubation at room temperature, are used to interpret the result.
PRECAUTION FOR USERS
1. For in-vitro diagnostic use only.
2. Handle all specimens as if they contain infectious agents. When the assay procedure is completed, dispose of specimens carefully after autoclaving for at least one hour. Alternatively, treat with a 0.5 or 1% solution of sodium hypochlorite for one hour before disposal.
3. Wear protective clothing (laboratory coats and disposable gloves) when assaying samples.
4. Do not eat, drink or smoke in areas where specimens and kit reagents are handled.
5. Avoid contact between hands and eyes or nose during specimen collection and testing.
Immunochromatographic rapid test card for qualitative detection of rotavirus antigen in feces
INTRODUCTION
Rotaviruses are the main cause of acute gastroenteritis, especially in children under the age of two years. Rotaviruses have been identified in almost 50% of the faeces of children with gastroenteritis, responsible for 600,000-800,000 death annually. Rotavirus infections occur frequently during the winter months. Gastroenteritis from enteric viruses can be mortal in risk populations such as children, the elderly or immunosuppressed individuals. Characteristic symptoms include vomiting, hydrodiarrhoea for between 3 and 8 days, high temperature and stomach pains. Rotaviruses transferred via the faecal-oral route are eliminated in large quantities into the intestine, so that hospital-borne infections from rotaviruses are regarded very seriously, particularly in baby stations and paediatric clinics, and are difficult to control. Early and reliable detection so that rotaviruses can be recognised and further infections avoided is therefore very important.
PRINCIPLE OF THE TEST
The Rotavirus Rapid Test Device employs red gold-conjugated monoclonal antibodies against antigen VP6 of group A of rotavirus, and solid-phase specific rotavirus antibodies. In this test the specimen is first treated with an extraction solution to extract rotavirus antigens from the faces. Following extraction, the only step required is to add the extract to the reaction device. As the sample flows through the test membrane, the colored particles migrate. In the case of a positive result the specific antibodies present on the membrane will capture the colored particles. Red color lines will be visible, depending upon the virus content of the sample. These lines, after 5-15 minutes of incubation at room temperature, are used to interpret the result.
PRECAUTION FOR USERS
1. For in-vitro diagnostic use only.
2. Handle all specimens as if they contain infectious agents. When the assay procedure is completed, dispose of specimens carefully after autoclaving for at least one hour. Alternatively, treat with a 0.5 or 1% solution of sodium hypochlorite for one hour before disposal.
3. Wear protective clothing (laboratory coats and disposable gloves) when assaying samples.
4. Do not eat, drink or smoke in areas where specimens and kit reagents are handled.
5. Avoid contact between hands and eyes or nose during specimen collection and testing.
SPECIMEN COLLECTION
Stool samples must be taken as soon as the symptoms appear. Viral particles decrease in number after one week, making the diagnosis more difficult. The samples can be stored in the refrigerator for 1 to 2 days. For longer storage they must be kept frozen at -20ºC. In this case, the sample should be totally thawed, and brought to room temperature and homogenised before testing.
STORAGE OF TEST KIT
The Rotavirus Rapid Test Device can be stored at any temperature between 4-30°C. Do not freeze. The stability of the kit under these storage conditions is 24 months. Use up the reagents as soon as possible after the kit is unpacked within 3 months.
ASSAY PROCEDURES
1. Allow all reagents to reach room temperature before use.
2. Place 0.5-1ml of extraction buffer in a properly marked testing tube.
3. Add a sample portion of approximately 5-6 mm size (25-100mg), with a swab, a wooden applicator or a bacteriology loop. Press the applicator to the tube and rotating it at the same time. For liquid or semi-solid stools add 100 microliters of stool using an appropriate pipette.
4. Vortex or stir to release the virus into diluent.
5. Add 3-4 drops to the sample well of the test device.
6. Incubate the test at room temperature and read the test after 5-15 minutes.
Stool samples must be taken as soon as the symptoms appear. Viral particles decrease in number after one week, making the diagnosis more difficult. The samples can be stored in the refrigerator for 1 to 2 days. For longer storage they must be kept frozen at -20ºC. In this case, the sample should be totally thawed, and brought to room temperature and homogenised before testing.
STORAGE OF TEST KIT
The Rotavirus Rapid Test Device can be stored at any temperature between 4-30°C. Do not freeze. The stability of the kit under these storage conditions is 24 months. Use up the reagents as soon as possible after the kit is unpacked within 3 months.
ASSAY PROCEDURES
1. Allow all reagents to reach room temperature before use.
2. Place 0.5-1ml of extraction buffer in a properly marked testing tube.
3. Add a sample portion of approximately 5-6 mm size (25-100mg), with a swab, a wooden applicator or a bacteriology loop. Press the applicator to the tube and rotating it at the same time. For liquid or semi-solid stools add 100 microliters of stool using an appropriate pipette.
4. Vortex or stir to release the virus into diluent.
5. Add 3-4 drops to the sample well of the test device.
6. Incubate the test at room temperature and read the test after 5-15 minutes.

INTERPRETATION OF RESULTS
Negative: One pink line appears in control line, showing the test has been carried out correctly.
Negative: One pink line appears in control line, showing the test has been carried out correctly.
There will be no line in test region
Positive: In addition to a pink colored control line, a distinct pink colored band will also appear in the test region.
Invalid: A total absence of color in both regions is an indication of procedure error and/or that the test reagent has
deteriorated. The test should be repeated using a new strip.
LIMITATIONS OF THE ASSAY
1. The test should be used only for the detection of rotavirus antigen in faecal samples.
2. The test is qualitative and no quantitative interpretation should be made with respect to the intensity of the positive line, when reporting the result
3. More than 500 samples were evaluated to assure the correct performance of the test. The correlation of the results with other techniques (ELISA) was excellent. However, interferences in the performance of the tests should not be excluded.
4. No cross-reactions with other viruses or substances were observed during the evaluation of the test. A negative result does not totally exclude a possible rotavirus infection.
Positive: In addition to a pink colored control line, a distinct pink colored band will also appear in the test region.
Invalid: A total absence of color in both regions is an indication of procedure error and/or that the test reagent has
deteriorated. The test should be repeated using a new strip.
LIMITATIONS OF THE ASSAY
1. The test should be used only for the detection of rotavirus antigen in faecal samples.
2. The test is qualitative and no quantitative interpretation should be made with respect to the intensity of the positive line, when reporting the result
3. More than 500 samples were evaluated to assure the correct performance of the test. The correlation of the results with other techniques (ELISA) was excellent. However, interferences in the performance of the tests should not be excluded.
4. No cross-reactions with other viruses or substances were observed during the evaluation of the test. A negative result does not totally exclude a possible rotavirus infection.
The significance of the results must be evaluated in relation to the patient’s clinical symptoms.
Company Profile
Biocare was founded by scientists and investers, to design, develop and manufacture monoclonal antibody, antigen and lateral flow colloidal gold tests and ELISA kits for infectious disease, cardiac marker, tumor marker, fertility, specific proteins. Like fecal calprotectin, pepsinogen I, pepsinogen II, AMH, vwf, GDH, NGAL, fFN, rotavirus, adenovirus, rota/adenovirus, H pylori antigen, faecal transferrin, fecal occult blood, lactoferrin, PROM IGFBP-1, troponin I, myoglobin, HBsAg, HIV, HCV, HAV, HCG, LH.
Biocare is one of the high quality IVD raw materials supplier in China. Over 100 kinds of paired antiobdy is available. All these monoclonals was developed by our scientits.
Because of the using of the REEL-TO-REEL production technology for lateral flow tests, the produced tests have a better batch to batch and intra-batch accuracy. This can improve the accuracy and efficiency of POC diagnostic testing to help increase patient safety.
Biocare is one of the high quality IVD raw materials supplier in China. Over 100 kinds of paired antiobdy is available. All these monoclonals was developed by our scientits.
Because of the using of the REEL-TO-REEL production technology for lateral flow tests, the produced tests have a better batch to batch and intra-batch accuracy. This can improve the accuracy and efficiency of POC diagnostic testing to help increase patient safety.







Certifications



FAQ
Q1. What is the advantage about your company?
A1. Our company was founded by professional scientists and investors. Have over twenty years experience for In Vitro Diagnostics Reagents to serve customer all over the world.
Q2. Why should I choose your products?
A2. Our products are high quality and low price.
Q3. The logo can be customized?
A3.Yes, We provide customized services.
Q4. How do you make our business long-term and good relationship?
A4. 1. We keep good quality and competitive price to ensure our customers‘benefit
2. We respect every customer as our friends and we sincerely do business.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices