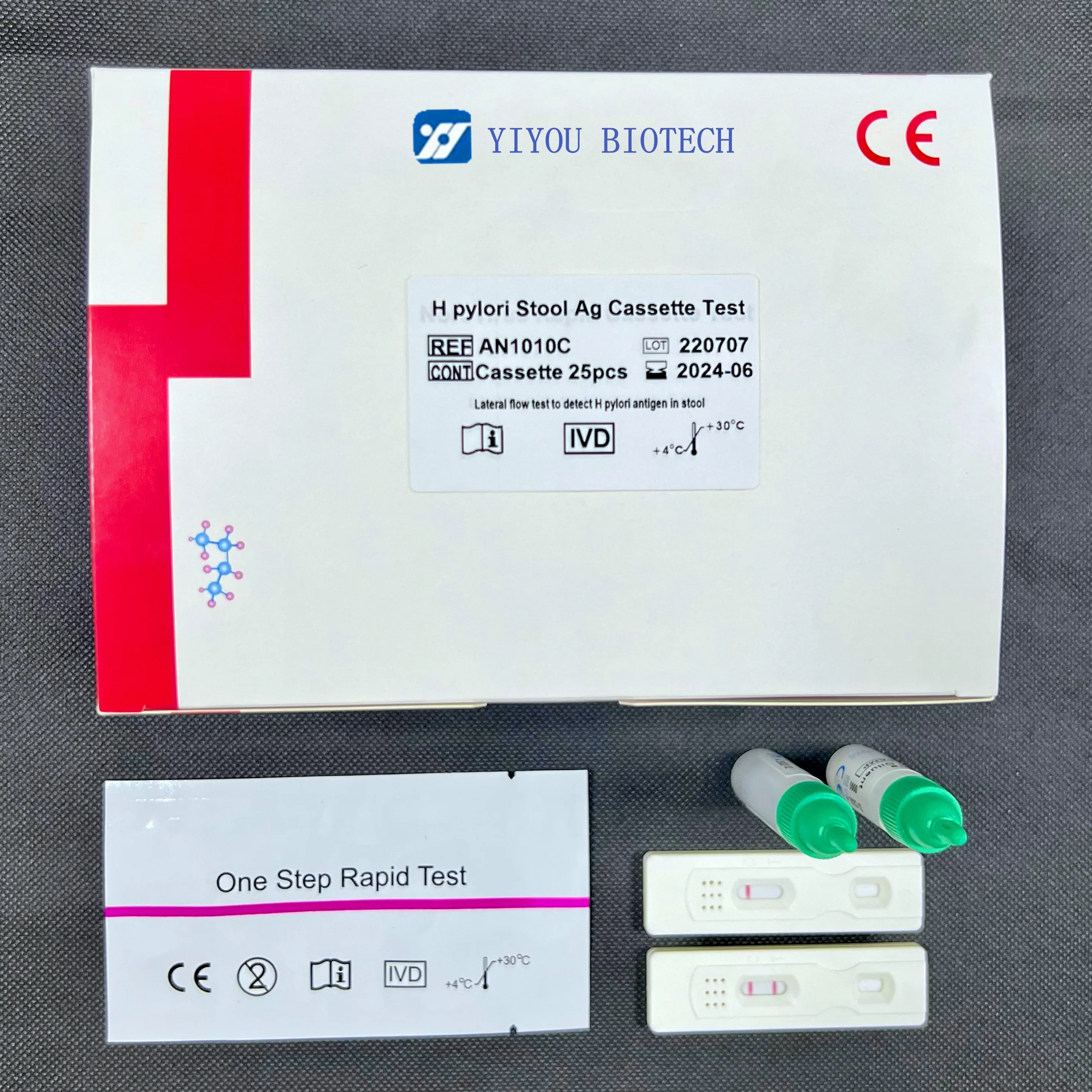
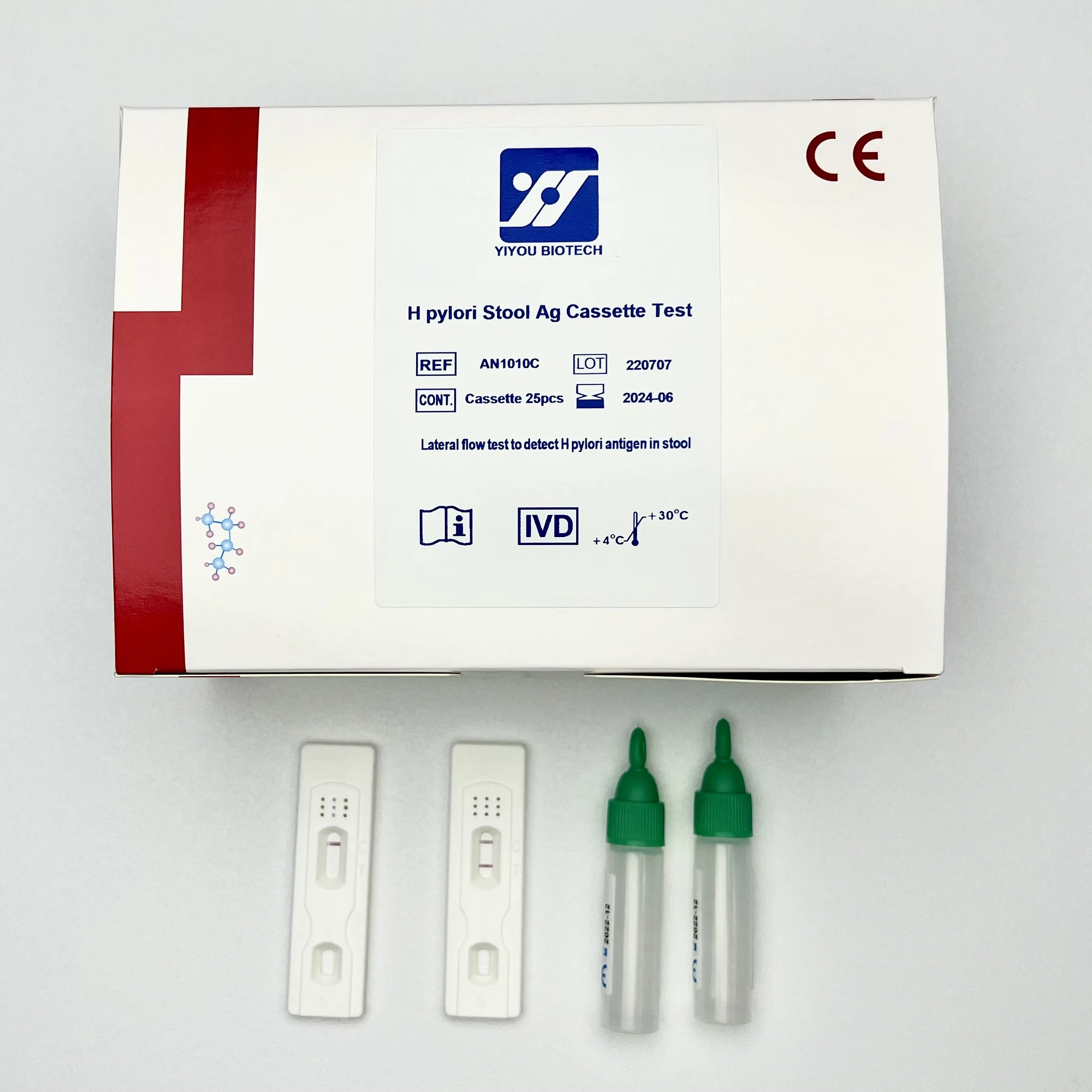
IVD Use Self Test H pylori Ag Rapid Test
- Category: Custom Chemical Services >>>
- Supplier: Biocare Diagnostics Ltd.
Share on (11000005851981):
Product Overview
Description
Specification
Brand Name | Biocare |
Product name | H pylori Ag Test |
Package Size | 25tests/kit, 10tests/kit |
Specimen | human feces |
Brand Name | Biocare or OEM |
Shelf Life | 24months |
Certificate | ISO13485 |
Storage | 4-30℃ |
Product Description
INTENDED USE
The H. pylori Antigen Test is an in vitro qualitative immunochromatographic assay for the rapid detection of Helicobacter pylori antigens in human stool specimen. The test results are intended to aid in the diagnosis of H. pylori infection, to monitor the effectiveness of therapeutic treatment and to confirm the eradication of H. pylori in peptic ulcer patients.
The H. pylori Antigen Test is an in vitro qualitative immunochromatographic assay for the rapid detection of Helicobacter pylori antigens in human stool specimen. The test results are intended to aid in the diagnosis of H. pylori infection, to monitor the effectiveness of therapeutic treatment and to confirm the eradication of H. pylori in peptic ulcer patients.
PRINCIPLE OF THE TEST
H. pylori Antigen Test is a sandwich solid phase immunochromatographic assay. To perform the test, an aliquot of diluted stool sample is added to the sample well of the test cassette. The sample flows through a label pad containing H. pylori antibody coupled to red-colored colloidal gold. If the sample contains H. pylori antigens, the antigen will bind to the antibody coated on the colloidal gold particles to form antigen-antibody-gold complexes. These complexes move on the nitrocellulose membrane by capillary action toward the test line region on which H. pylori specific antibodies are immobilized. As the complexes reach the test line, they will bind to the antibody on the membrane in the form of a line. A second red control line will always appear in the result window to indicate that the test has been correctly performed and the test device functions properly. If H. pylori antigen is not present or lower than the detection limit of the test, only the control line will be visible. If the control line dose not developed, the test is invalid.
H. pylori Antigen Test is a sandwich solid phase immunochromatographic assay. To perform the test, an aliquot of diluted stool sample is added to the sample well of the test cassette. The sample flows through a label pad containing H. pylori antibody coupled to red-colored colloidal gold. If the sample contains H. pylori antigens, the antigen will bind to the antibody coated on the colloidal gold particles to form antigen-antibody-gold complexes. These complexes move on the nitrocellulose membrane by capillary action toward the test line region on which H. pylori specific antibodies are immobilized. As the complexes reach the test line, they will bind to the antibody on the membrane in the form of a line. A second red control line will always appear in the result window to indicate that the test has been correctly performed and the test device functions properly. If H. pylori antigen is not present or lower than the detection limit of the test, only the control line will be visible. If the control line dose not developed, the test is invalid.
PRECAUTION FOR USERS
1. For in-vitro diagnostic use only.
2. Must not use kit beyond the expiration date.
3. Do not mix components from kits with different lot number.
4. Avoid microbial contamination of reagents.
5. Do not pipet reagent by mouth and no smoking or eating while performing assays.
6. Wear gloves during the whole process and avoid reagents or specimen spilling-out.
7. Wipe up the spills using 5% hypochlorite solution.
8. Decontaminate all liquids or solid wastes before deposing.
1. For in-vitro diagnostic use only.
2. Must not use kit beyond the expiration date.
3. Do not mix components from kits with different lot number.
4. Avoid microbial contamination of reagents.
5. Do not pipet reagent by mouth and no smoking or eating while performing assays.
6. Wear gloves during the whole process and avoid reagents or specimen spilling-out.
7. Wipe up the spills using 5% hypochlorite solution.
8. Decontaminate all liquids or solid wastes before deposing.
SPECIMEN COLLECTION
Stool specimens should be collected in containers that do not contain media, preservatives, animal serum or detergents as any of these additives may interfere with the HP Ag Test. Specimens may be stored at 2-8°C for 3 days without interfering with the assay performance. For long-term storage of specimens, -20°C or colder is recommended. Repeated freezing and thawing of specimens is not recommended and may cause erroneous results. Do not store specimens in self-defrosting freezers.
STORAGE OF TEST KIT
The HP Ag Test can be stored at any temperature between 4-30°C. Do not freeze. The stability of the kit under these storage conditions is 24 months. Use up the reagents as soon as possible after the kit is unpacked within 3 months.
ASSAY PROCEDURES
1. Allow all reagents to reach room temperature before use.
2. Place 0.5-1ml of extraction buffer in a properly marked testing tube.
3. Add a sample portion of approximately 5-6 mm size (25-100mg), with a swab, a wooden applicator or a bacteriology loop. Press the applicator to the tube and rotating it at the same time. For liquid or semi-solid stools add 100 microliters of stool using an appropriate pipette.
4. Vortex or stir to release the virus into diluent.
5. Add 3-4 drops to the sample well of the test device.
6. Incubate the test at room temperature and read the test after 5-15 minutes.
INTERPRETATION OF RESULTS
Stool specimens should be collected in containers that do not contain media, preservatives, animal serum or detergents as any of these additives may interfere with the HP Ag Test. Specimens may be stored at 2-8°C for 3 days without interfering with the assay performance. For long-term storage of specimens, -20°C or colder is recommended. Repeated freezing and thawing of specimens is not recommended and may cause erroneous results. Do not store specimens in self-defrosting freezers.
STORAGE OF TEST KIT
The HP Ag Test can be stored at any temperature between 4-30°C. Do not freeze. The stability of the kit under these storage conditions is 24 months. Use up the reagents as soon as possible after the kit is unpacked within 3 months.
ASSAY PROCEDURES
1. Allow all reagents to reach room temperature before use.
2. Place 0.5-1ml of extraction buffer in a properly marked testing tube.
3. Add a sample portion of approximately 5-6 mm size (25-100mg), with a swab, a wooden applicator or a bacteriology loop. Press the applicator to the tube and rotating it at the same time. For liquid or semi-solid stools add 100 microliters of stool using an appropriate pipette.
4. Vortex or stir to release the virus into diluent.
5. Add 3-4 drops to the sample well of the test device.
6. Incubate the test at room temperature and read the test after 5-15 minutes.
INTERPRETATION OF RESULTS

INTERPRETATION OF RESULTS
Negative: One pink line appears in control line, showing the test has been carried out correctly.
Negative: One pink line appears in control line, showing the test has been carried out correctly.
There will be no line in test region.
Positive: In addition to a pink colored control line, a distinct pink colored band will also appear in the test region.
Invalid: A total absence of color in both regions is an indication of procedure error and/or that the test reagent has
deteriorated. The test should be repeated using a new strip.
Positive: In addition to a pink colored control line, a distinct pink colored band will also appear in the test region.
Invalid: A total absence of color in both regions is an indication of procedure error and/or that the test reagent has
deteriorated. The test should be repeated using a new strip.
Company Profile
Biocare was founded by scientists and investers, to design, develop and manufacture monoclonal antibody, antigen and lateral flow colloidal gold tests and ELISA kits for infectious disease, cardiac marker, tumor marker, fertility, specific proteins. Like fecal calprotectin, pepsinogen I, pepsinogen II, AMH, vwf, GDH, NGAL, fFN, rotavirus, adenovirus, rota/adenovirus, H pylori antigen, faecal transferrin, fecal occult blood, lactoferrin, PROM IGFBP-1, troponin I, myoglobin, HBsAg, HIV, HCV, HAV, HCG, LH.
Biocare is one of the high quality IVD raw materials supplier in China. Over 100 kinds of paired antiobdy is available. All these monoclonals was developed by our scientits.
Because of the using of the REEL-TO-REEL production technology for lateral flow tests, the produced tests have a better batch to batch and intra-batch accuracy. This can improve the accuracy and efficiency of POC diagnostic testing to help increase patient safety.
Biocare is one of the high quality IVD raw materials supplier in China. Over 100 kinds of paired antiobdy is available. All these monoclonals was developed by our scientits.
Because of the using of the REEL-TO-REEL production technology for lateral flow tests, the produced tests have a better batch to batch and intra-batch accuracy. This can improve the accuracy and efficiency of POC diagnostic testing to help increase patient safety.







Certifications



FAQ
Q1. What is the advantage about your company?
A1. Our company was founded by professional scientists and investors. Have over twenty years experience for In Vitro Diagnostics Reagents to serve customer all over the world.
Q2. Why should I choose your products?
A2. Our products are high quality and low price.
Q3. The logo can be customized?
A3.Yes, We provide customized services.
Q4. How do you make our business long-term and good relationship?
A4. 1. We keep good quality and competitive price to ensure our customers‘benefit
2. We respect every customer as our friends and we sincerely do business.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices