ProteinIso Ni NTA Resin His Tag Protein
- Category: Other Chemicals >>>
- Supplier: TransGen Biotech Co. Ltd.
Share on (1424289937):
Product Overview
Description
ProteinIso Ni-NTA Resin
ProteinIsoTM Ni-NTA Resin is used for one-step purification of Histagged proteins. The His-tagged proteins bind to Ni2+ cations, which are immobilized on the Ni-NTA resin by 4 metal-chelating sites. After unbound proteins are washed away, the target proteins are recovered by gradient elution. It is suitable for both native and denatured protein purification.
| Cat. No. | ||
| Specification | DP101-01 | 5 ml |
| DP101-02 | 25 ml | |
| Storage: | at 2~8oC (20% ethanol) for two years | |
| Specification | Samples should be centrifuged and filtrated with 0.45 μm filter before loading. | |
Equilibration Buffer for soluble protein: 300 mM NaCl, 50 mM sodium phosphate buffer, 10 mM imidazole, 10 mM Tris-Cl pH 8.0 | ||
| Equilibration Buffer for inclusion body: 6 M GuHCl, 100 mM sodium phosphate buffer; or 8 M urea, 100 mM sodium phosphate buffer, 10 mM Tris-Cl pH 8.0 | ||
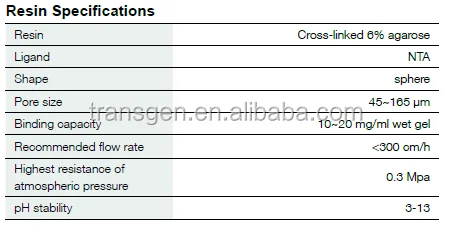
Resin Specifications
| References | Specifications |
| Matrix | 6% cross-linked agarose |
| Ligand | NTA |
| Shape | sphere |
| Pore size | 45-165 μm |
| Binding capacity | 10-20 mg proteins/ml wet gel |
| Recommended flow rate | <300 cm/h |
| Highest resistance of atmospheric pressure | 0.3 Mpa |
| pH stability | 3-13 |
Procedures
1. Prepare Ni-NTA purification column
(1) Resuspend the Ni-NTA resin in its bottle by inverting.
(2) Transfer the resin into a purification column. Allow the resin to settle.
(3) Equilibrate the column with 5~10 bed volume of equilibration buffer.
2. Prepare samples
To avoid blocking column, samples should be centrifuged and filtrated before loading.
3. Load samples and wash
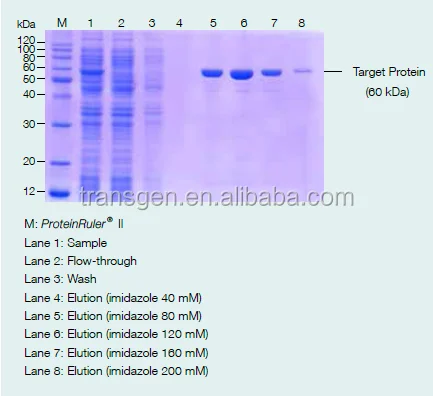
Load samples and wash with 5~10 bed volume of equilibration buffer and collect the flow-through in one tube.
4. Elute
Elute target protein with different concentration imidazole or different pH equilibration buffer.
5. Resin Regeneration
(1) 2 bed volume of 6 M GuHCl, 0.2 M acetic acid
(2) 5 bed volume of deionized water
(3) 3 bed volume of 2% SDS
(4) 1 bed volume of 25% ethanol
(5) 1 bed volume of 50% ethanol
(6) 1 bed volume of 75% ethanol
(7) 5 bed volume of 100% ethanol
(8) 1 bed volume of 75% ethanol
(9) 1 bed volume of 50% ethanol
(10) 1 bed volume of 25% ethanol
(11) 1 bed volume of deionized water
(12) 5 bed volume of 100 mM EDTA, pH 8.0
(13) 10 bed volume of deionized water
(14) 5 bed volume of 100 mM NiSO4



CITATIONS
Cao H, 2013. FCHSD1 and FCHSD2 Are Expressed in Hair Cell Stereocilia and Cuticular Plate and Regulate Actin Polymerization In Vitro. 8(2):e56516. PLoS One. IF=3.73. PMID: 23437151.
Wang L, et al. 2011. Cloning of exoinulinase gene from Penicillium janthinellum strain B01 and its high-level expression in Pichia pastoris. J Appl Microbiol. IF=2.196. PMID: 21895898.
Huang H, 2013. Cloning, expression and characterization of a phosphoglucomutase/phosphomannomutase from sphingan-producing Sphingomonas sanxanigenens. 35(8):1265-70. Biotechnol Lett. IF=1.853. PMID: 23546942.
We Recommend
Cheap and best Caluanie Muelear Oxidize Parteurized
CAS 143-18-0 Catalyst Emulsifier Potassium Oleate
Good price Boron Nitride Nanosheets BN Nanosheets
China Factory of Trinexapac-ethyl CAS: 95266-40-3 with high-quality
New Arrivals
New products from manufacturers at wholesale prices