Reliable Reputation Voyage Toxo IgG Elisa Kit
- Category: >>>
- Supplier: Nantong Voyage International Trade Co. Ltd. [Jiangsu China]Nantong
Share on (1600072183283):
Product Overview
Description


The basic enzyme-linked immunosorbent assay (ELISA), or enzyme immunoassay (EIA), is distinguished from other antibody-based assays because separation of specific and non-specific interactions occurs via serial binding to a solid surface, usually a polystyrene multiwell plate, and because quantitative results can be achieved.
The ELISA procedure results in a colored end product which correlates to the amount of analyte present in the original sample.
ELISAs are quick and simple to carry out, and since they are designed to rapidly handle a large number of samples in parallel, they are a very popular choice for the evaluation of various research and diagnostic targets. Figure 1 shows a typical ELISA result.
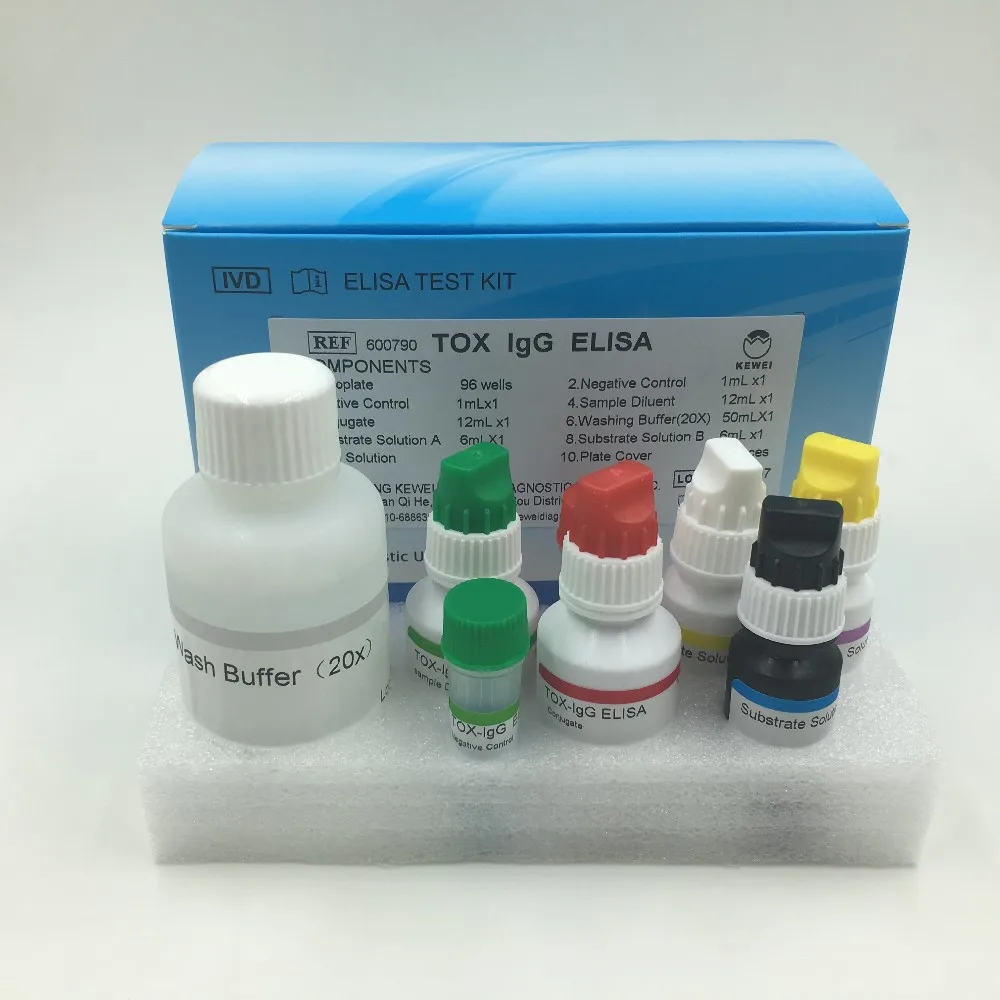
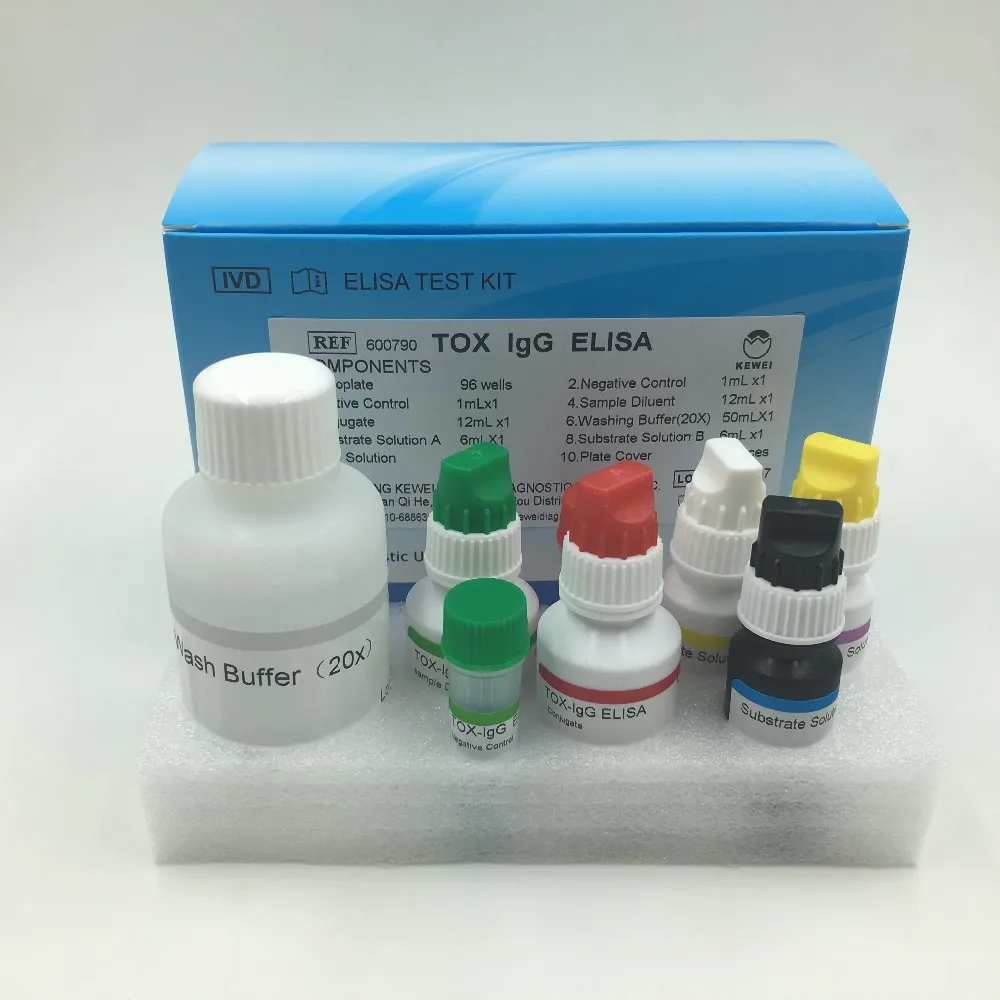
Composition of kit | ||
Microplate coated with Toxoplasma antigen | 96 holes | |
Enzyme labeled anti human IgG antibody | 6.5ml | |
Positive control serum | 0.8ml | |
Critical positive control serum | 0.8ml | |
Negative control serum | 0.8ml | |
Developer A | 6.5ml | |
Developer B | 6.5ml | |
Stop fluid | 6.5ml | |
Concentrated detergent (10 times) | 50ml | |
Sample diluent | 52mlx2 | |


Our Factory
voyage medical co.,Itd is a high-tech bio-technology company and our factory was found in 1999.at present, we reagrd human health as our own ideal, and continues to study for the public to make more accurate and faster and more convenient products to facilitate the disease diagnosis.





Advantages
1. Popular sell at home and abroad.






We Recommend
New Arrivals
New products from manufacturers at wholesale prices