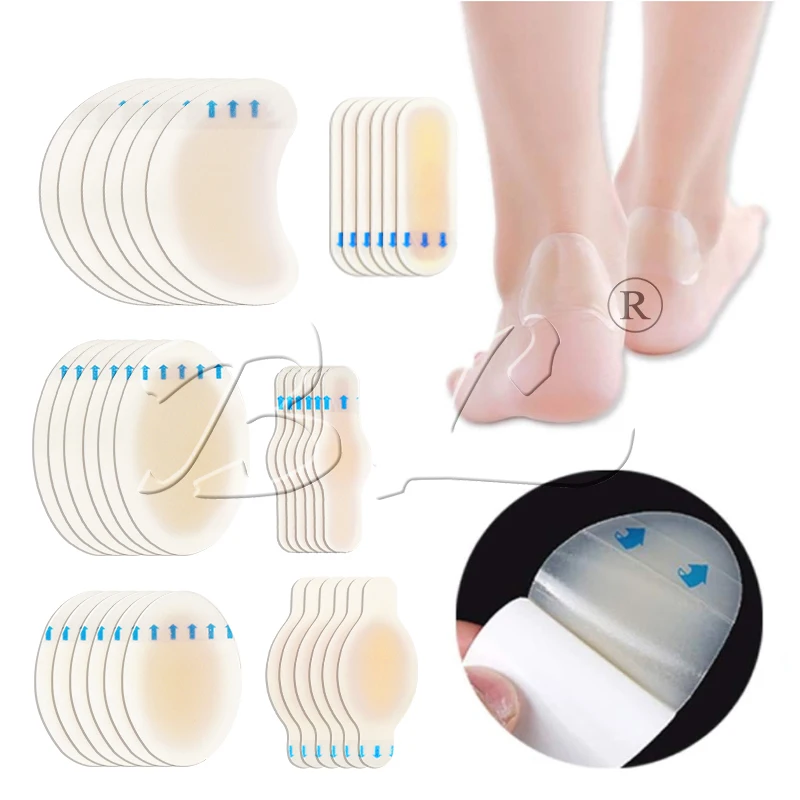
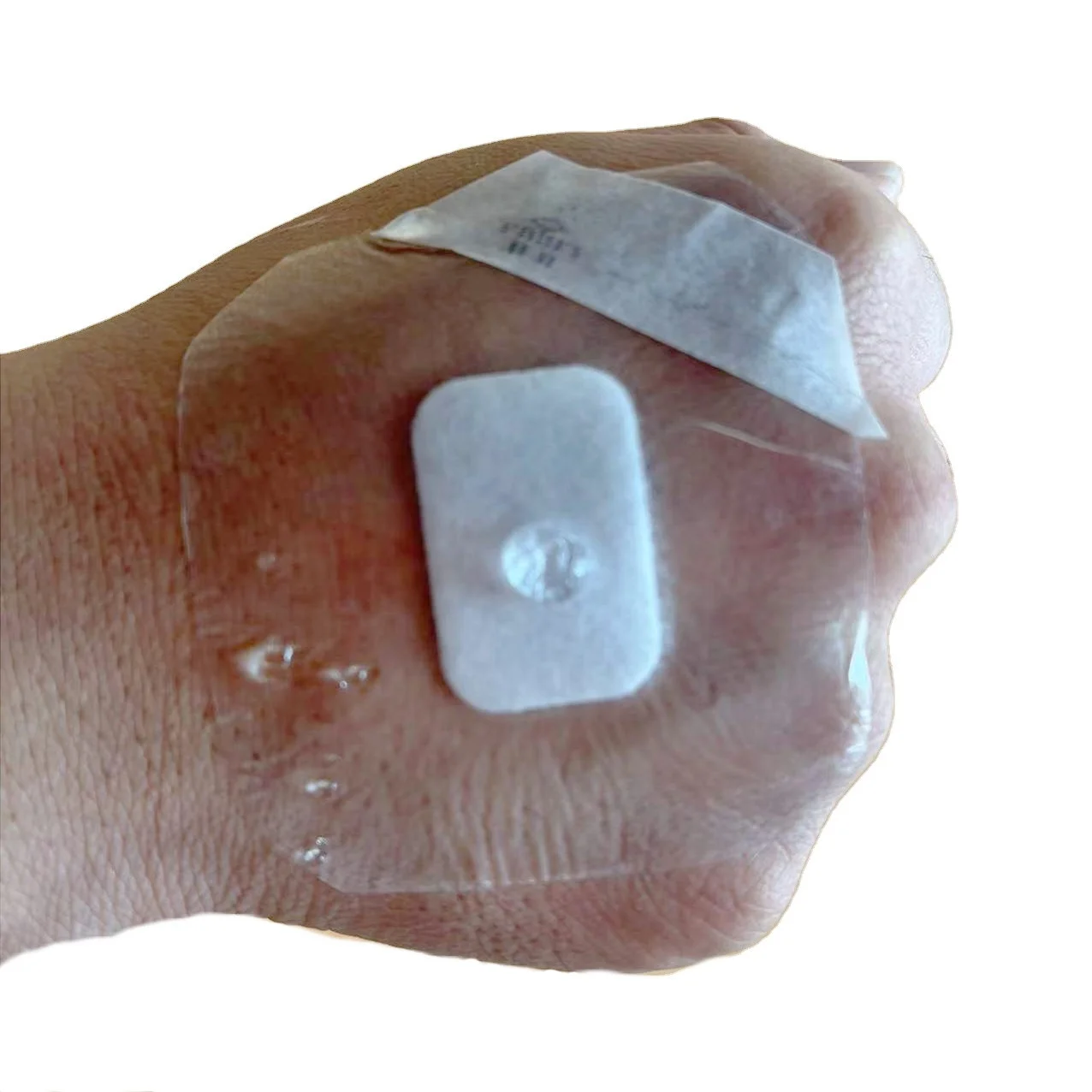
Adhesive Transparent Island Dressing for Wound Care
- Category: >>>
- Supplier: Jiangsu Newvalue Medical Products Co. Ltd.
Share on (1600178902822):
Product Overview
Description
Product Description
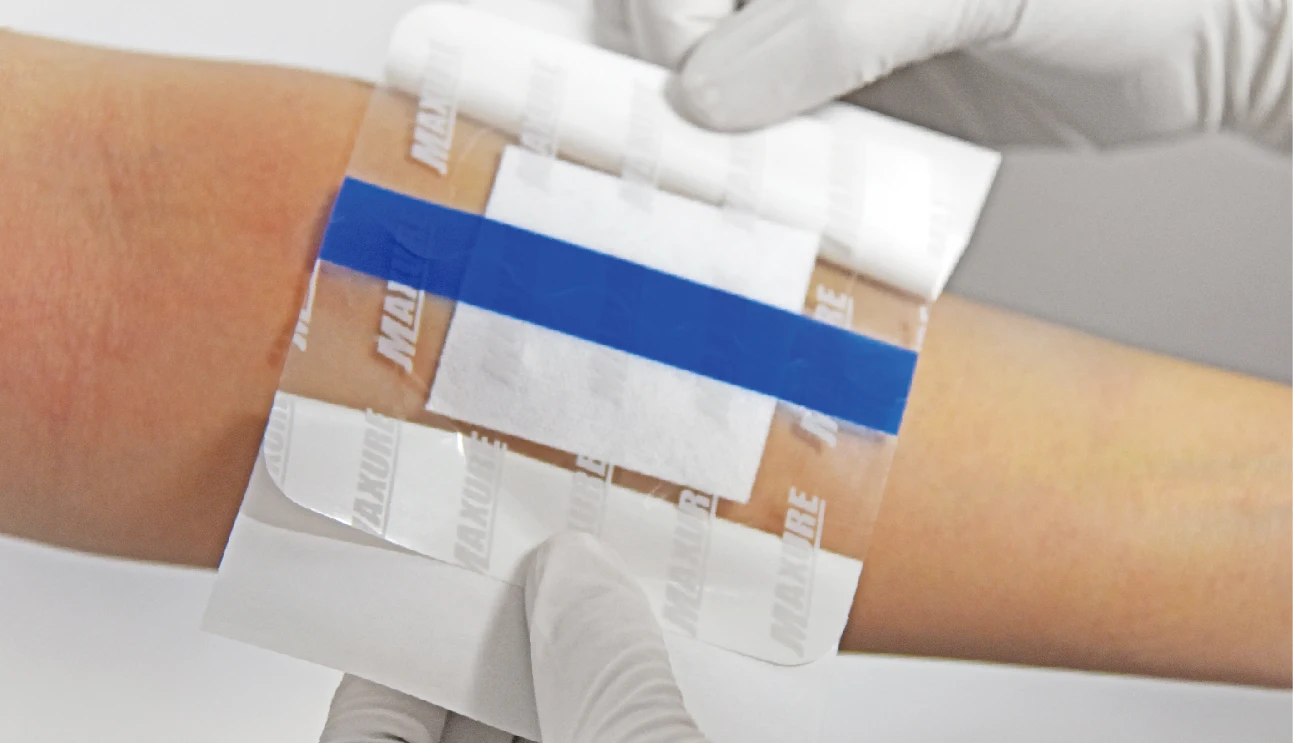
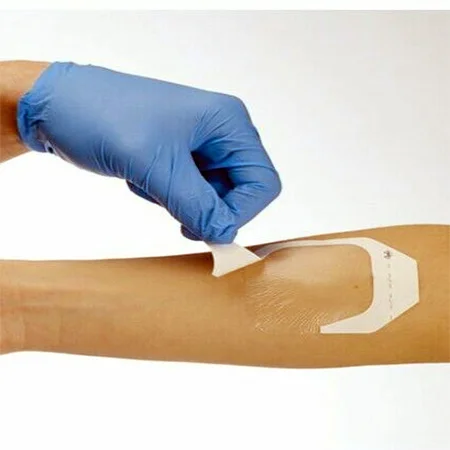
MAXURE Transparent Wound Dressing
Post-Operative Dressing • easy • safe • stable
• Hypoallergenic and Without Natural Rubber Latex
• Gentle to skin and breathable
• Soft and comfortable
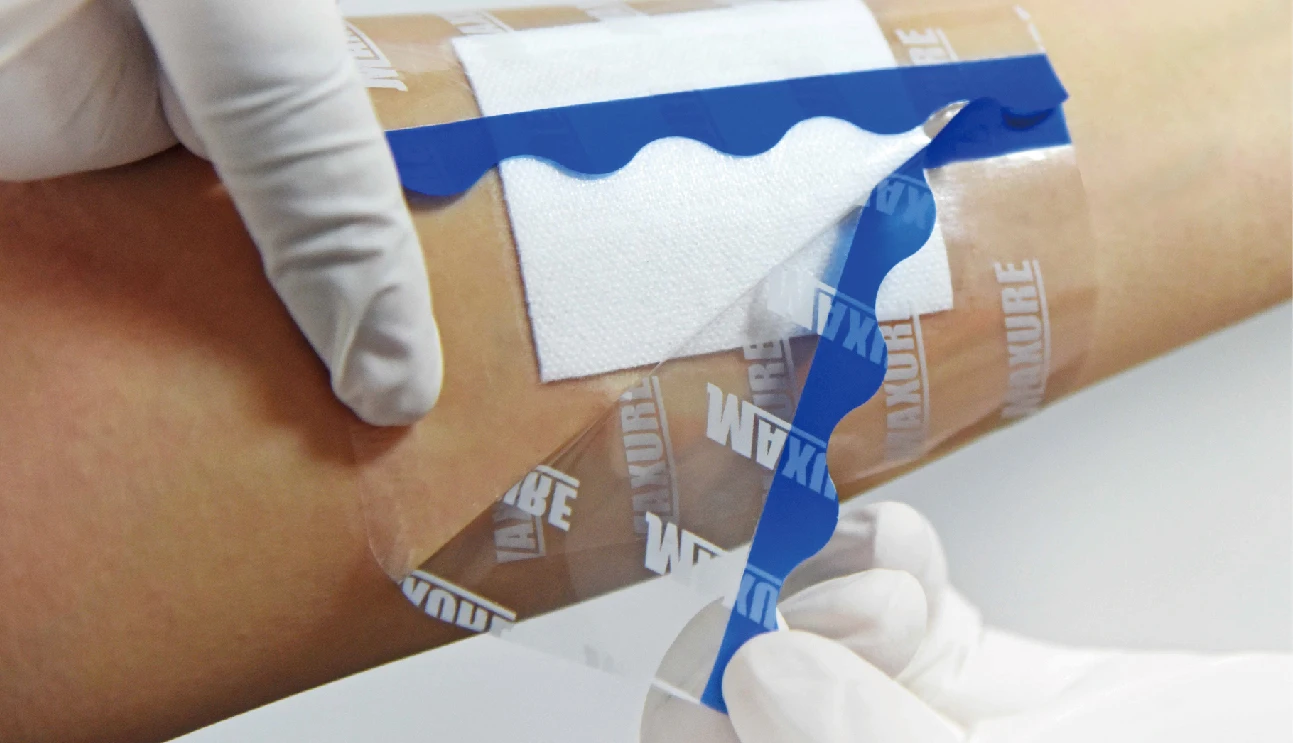
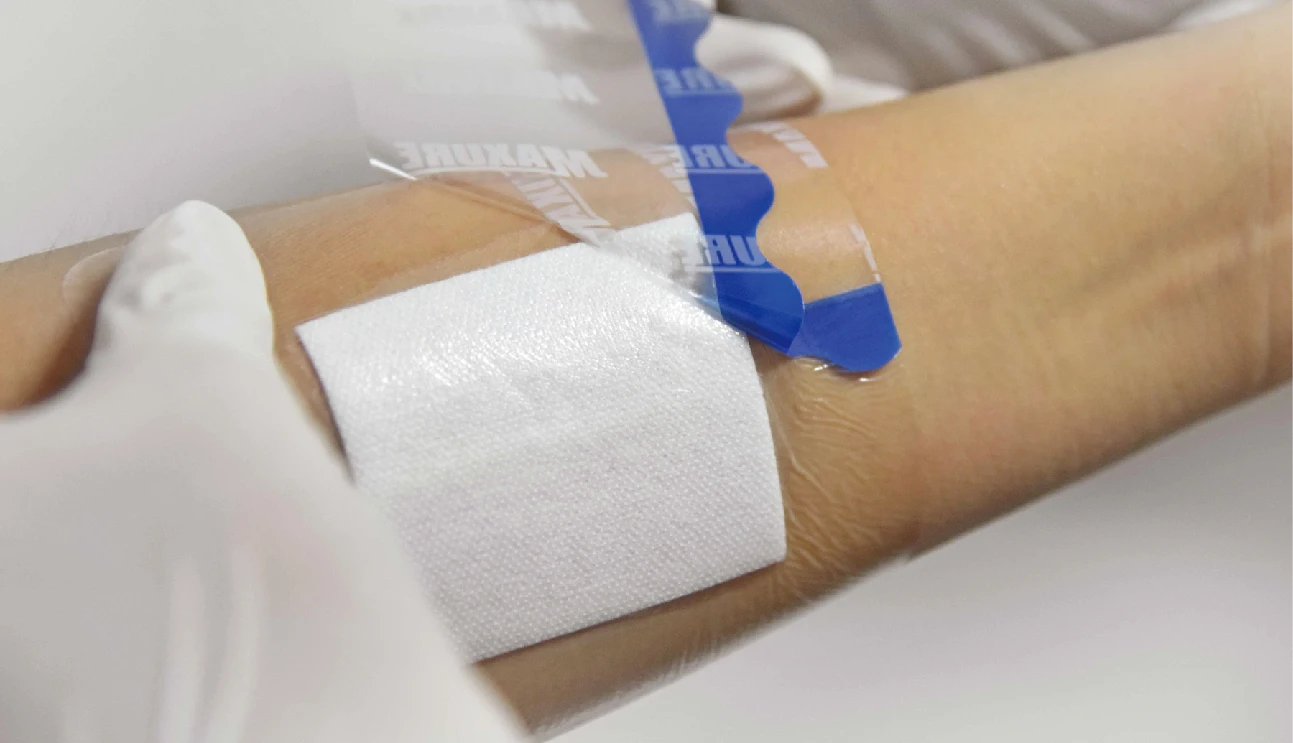
• Lifting design available, easy to apply
• Easy to apply with user-friendly design for lifting
• Elastic and flexible for moving and bending body parts
• Good tensile strength and durable
• Gentle to skin and breathable
• Soft and comfortable
• Lifting design available, easy to apply
• Easy to apply with user-friendly design for lifting
• Elastic and flexible for moving and bending body parts
• Good tensile strength and durable
Specification
Product Name | Transparent Wound Dressing |
Place of Origin | China |
Brand Name | MAXURE |
Model Number | 621020F |
Material | Polyurethane, Medical Grade Adhesive, Wound Pad |
Properties | PU Film Dressing • easy • safe • stable |
Size | 10*20cm |
Shelf Life | 3 years |
Certificate | CE, ISO13485, USA |
Sterile | Yes |
Package | Individual Medical Bag |
Sample | Avaliable |
MOQ | 10000pcs |
Delivery time | 4-6 weeks |
Packing & Delivery
1 pc/pouch, 25 pcs/box, 4 boxes/case


Company Profile
• New Value was founded in 2010 by our core team who used to work for Fortune 500 company in different functions including sales, technical and R&D. Given our background, we focus continuously on material development and innovation for medical applications such as advanced wound care and hemostasis.
• Our fully owned manufacturing facility is located in China Medical City at Taizhou, Jiangsu province. The facility is equipped with two clean rooms (Class 100K and 10K each) as well as control-room level material work shop.
• With regard to business model, we supply both roll-form medical tape materials to manufacturers and finished medical device/consumer products in our customers’ brand.
• Our goal is to offer our customers premium products with top-of-the-line performance and quality with reasonable price to better serve their needs as well as their customers, patients in the hospitals or retail consumers.
• Our fully owned manufacturing facility is located in China Medical City at Taizhou, Jiangsu province. The facility is equipped with two clean rooms (Class 100K and 10K each) as well as control-room level material work shop.
• With regard to business model, we supply both roll-form medical tape materials to manufacturers and finished medical device/consumer products in our customers’ brand.
• Our goal is to offer our customers premium products with top-of-the-line performance and quality with reasonable price to better serve their needs as well as their customers, patients in the hospitals or retail consumers.



FAQ
Q: Are you a trading company or a manufacturer?
A: NewValue is a manufacturer. Our manufacturing facility is based in Taizhou city, Jiangsu province. We also have an office in Pudong district, Shanghai.
Q: Is your facility certified with any quality system?
A: Yes our facility is ISO-13485 2016 certified, and is also compliant with EU, US and China requirements.
Q: Do you have a clean room in your facility?
A: Yes, we have actually two clean rooms. We have a class 100K clean room (800m2) on the second floor and a class 10K clean room(600m2) on the third floor.
Q: What registration have you obtained for your hydrocolloid?
A: Our hydrocolloid is CE marked as a Class 2B medical device, registered in China as a Class 3 medical device (free sales certificate available). It is also registered in US, Japan, Korea, Indonesia and a few other countries.
Q: Do you make your own hydrocolloid or do you buy material in roll form to convert?
A: We developed our own hydrocolloid formulation and process. We mix, extrude and convert our hydrocolloid dressing all in-house.
Q: Are you already selling your hydrocolloid internationally?
A: Yes, we are already selling our hydrocolloid to customers in US, UK, Germany, Japan, Korea and a few other countries across the world.
A: NewValue is a manufacturer. Our manufacturing facility is based in Taizhou city, Jiangsu province. We also have an office in Pudong district, Shanghai.
Q: Is your facility certified with any quality system?
A: Yes our facility is ISO-13485 2016 certified, and is also compliant with EU, US and China requirements.
Q: Do you have a clean room in your facility?
A: Yes, we have actually two clean rooms. We have a class 100K clean room (800m2) on the second floor and a class 10K clean room(600m2) on the third floor.
Q: What registration have you obtained for your hydrocolloid?
A: Our hydrocolloid is CE marked as a Class 2B medical device, registered in China as a Class 3 medical device (free sales certificate available). It is also registered in US, Japan, Korea, Indonesia and a few other countries.
Q: Do you make your own hydrocolloid or do you buy material in roll form to convert?
A: We developed our own hydrocolloid formulation and process. We mix, extrude and convert our hydrocolloid dressing all in-house.
Q: Are you already selling your hydrocolloid internationally?
A: Yes, we are already selling our hydrocolloid to customers in US, UK, Germany, Japan, Korea and a few other countries across the world.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices