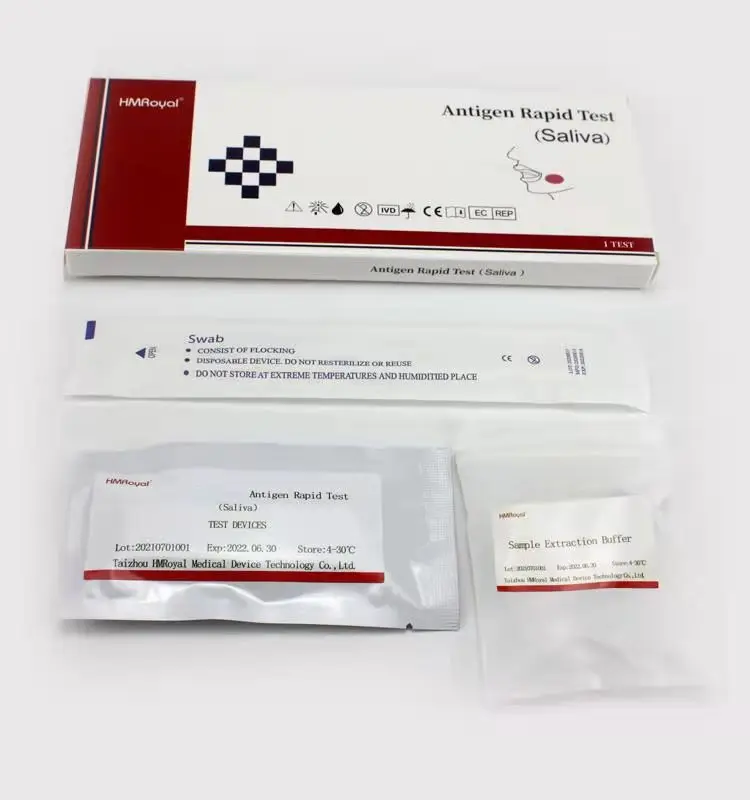
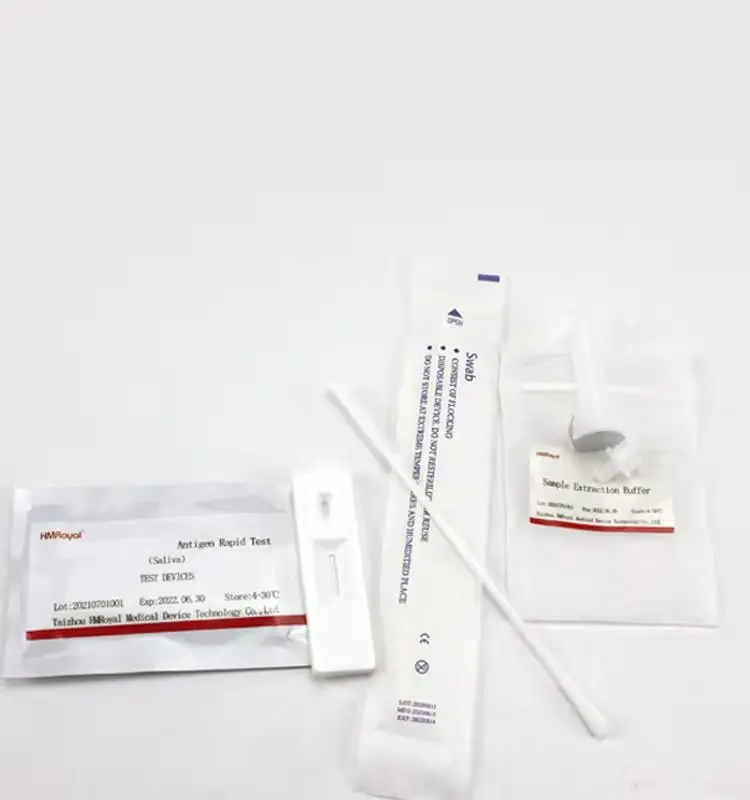
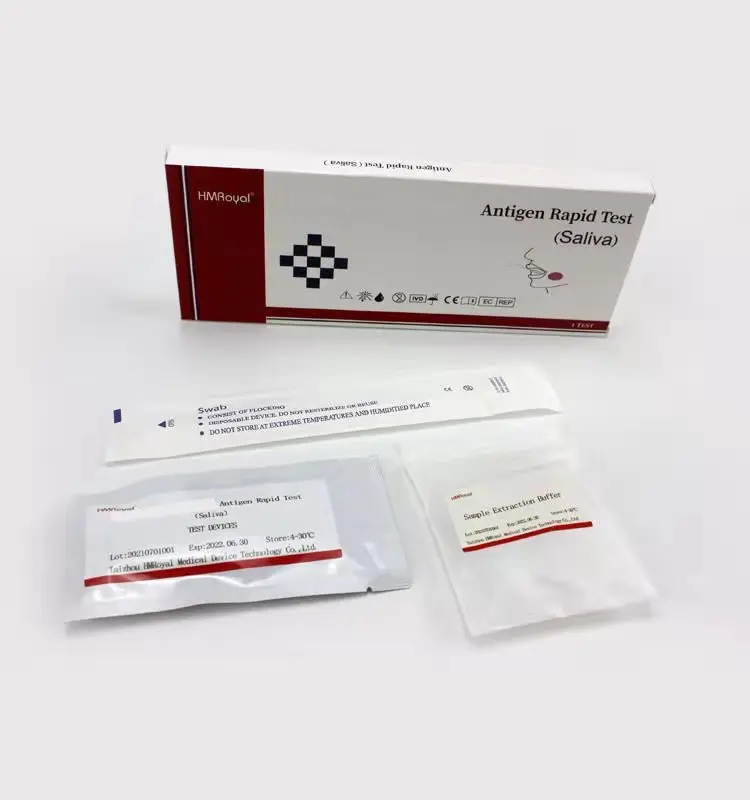
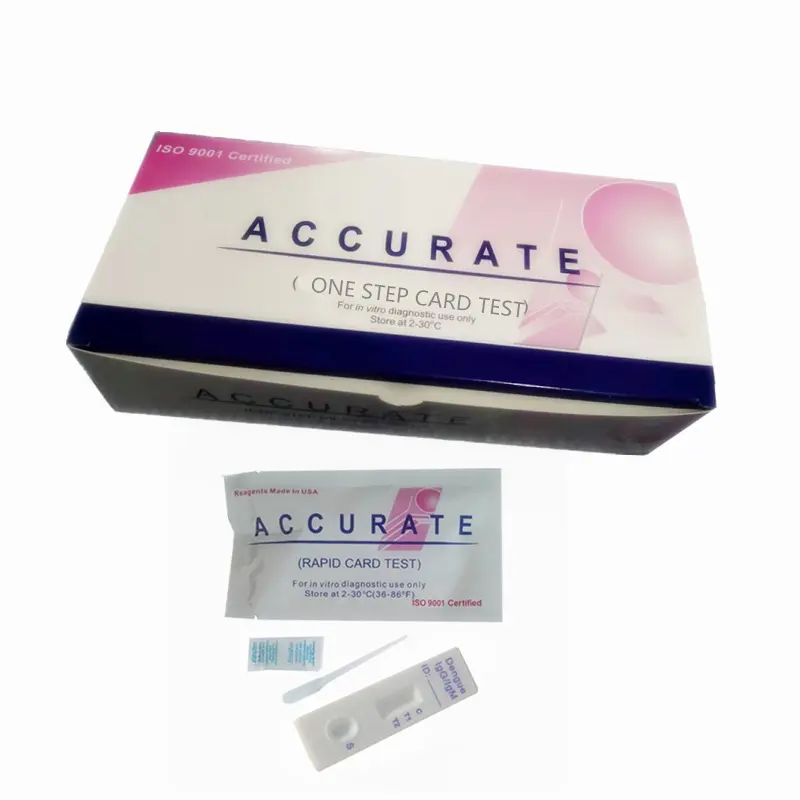
Домашнее тестирование слюны и носовых тампонов, антиген, быстрый диагностический тест, комплект для быстрого тестирования антигена, слюна
- Category: Pathological Analysis Equipments >>>
- Supplier: Taizhou Hmroyal Medical Device Technology Co. Ltd.
Share on (1600334308177):
Product Overview
Description

Product Paramenters

Product name | Antigen rapid test kit |
Specimen | Throat swab,Nasal swab,Sputum |
Format | Cassette |
Size | Box Size:12.5*6.5*1.5/Box Size:26*12*6.5 |
Shelf life and Storage | 18 months at room temperature 4- 30 degrees |
Components | Antigen Test Cassette,Sample extraction buffer,Disposable sampling swab,Biohazard specimen bag |
Reaction Time | 10-15minutes |
Using place | home use or hospital/clinics |
COMPONENTS
1.Antigen Test device
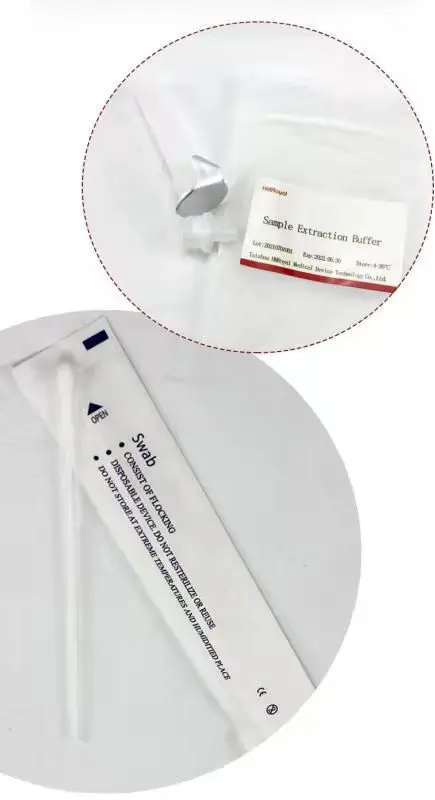
2.Sample extraction buffer
3.Disposable virus sampling swab
4.Instruction for use
Regents from different Lot could not be used in a test in case of error results
2.Sample extraction buffer
3.Disposable virus sampling swab
4.Instruction for use
Regents from different Lot could not be used in a test in case of error results

Specimen Requirements

● Insert the swab into the mouth cavity and place the swab head between upper and lower molar teeth. Let patient slightly bite the swab head for 10 seconds for saliva absorption.
● After saliva collection, gently take out the swab, put the swab with the sample into the Sample Extraction Bufferfor processing.

● The swab after sampling is soaked below the liquid level ofthe sample extraction buffer. Rotate and press 3 times. The swab soaking time is not less than 15 seconds.
● The swab head is pressed, then take outthe swab and tighten the sampling tube.

The treated sample should be tested within 30 minutes.
TEST PROCEDURE
1. Place the test device, sample extraction.buffer at room temperature for 15-30 minutes, and equilibrate to room temperature (10~30℃)

2. Open the aluminum foil pouch ofthe test cassette, place the test cassette on a flat surface.

3. Add 2-3 drops of the treated sample into th sample well of the test cassette. (In case of chromatographic abnormalities, add an extra of 1- 2 drops of the treated sample accordingly). Incubate at 10 30℃ for 15 minutes.

The result obtained after 30 minutes is invalid.
Disposal The Sample And Clean-up



Interpretation Of Result
Positive:
Two color bands appear in the observation window, thatis, a red or magenta line appears atthe position ofthe quality control line (C line) and 'the detection line (T line) (as shown in result 1), indicating the test result of antigens in the sample is positive.
Negative:
A red or magenta line appears atthe position ofthe quality control line (C line) in the observation window, and no line appears atthe position "ofthe test line (T line) (as shown in the result 2), indicating the test result ofthe antigens in the sample is negative or the concentration is below the limit of detection ofthe kit.
Nvalid:
No line appears in the position ofthe quality control line (line C) in the observation window (as shown in result 3), indicating thatthe testis invalid, and the sample should be recollect and retested

Principle of the Procedure
This kits based on the colloidal gold immunochromatographic technology,and uses the double antibody sandwich method to detect N protein of antigen in human saliva swab samples. The detection line (T line) of the antigen test cassette was coated with antibody, and the quality control line(Cline) was coated with sheep anti-mouse antibody. During the test,the sample is dropped into the test cassette and the liquid is chromatographed upward under the capillary effect. The antigen in the sample first binds to the colloidal gold-labelled antibody to forma solid phase antibody antigen-labelled antibodycolloidal gold complex at the Tline position, and form a solid phasesheep anti-mouse-labelled antibody-colloidal gold complex was formed at the C line position. After the testis completed, observe the colloidalgold color reaction of T line and Cline to determine results of antigen in human saliva swab samples
Storage and Stability
4~30℃ storage in dark and dry, the validity is 12 months. Freezing or use after expiration is prohibited.Stability after first open: 30 minutes at 10℃~30℃, RH 50%~60% Manufacture date and Expiry date are shown on the packaging label.
Company Profile
Taizhou HMRoyal Medical Device Technology Co., Ltd. is located in Taizhou pharmaceutical High-tech Industrial Development Zone, the pharmaceutical city of China. The park is jointly organized by the Ministry of science and technology of the people's Republic of China, the national health and Family Planning Commission, the State Food and drug administration, the China Pharmaceutical Administration and the Jiangsu provincial government. It is the first high-tech development zone with life science as the industrial theme. The company is introduced by the government, and now has 20000 square meters of modern workshop, 100000 dust-free
workshop and UV conversion room, with an annual output of 200million adult masks, 45 million children masks and 200million FFP2 and kn95 masks. In the key stage of the work of epidemic prevention in China, it has made important contributions to the work of epidemic prevention.
Our company mainly produces: disposable medical mask, surgical mask, dust mask,FFP2, FFP3, KN95 mask, PM2.5 mask, nano mask, fresh air mask, kids mask ;antigen self-test kit. Medical protective clothing, civil protective clothing, disposable isolation clothing, virus protective clothing, brush hand suit, clean clothing, hand washing clothes, operation clothes, surgical clothes, surgical clothes, doctor nurse clothes, sterile caps, surgical caps, shoe covers, medical gloves, medical rubber gloves, sterile film hand covers, sterile drug giving fingersets, inspection gloves, latex gloves, PE gloves, PVC gloves, Nitrile gloves, Isolation protection: medical
Our company mainly produces: disposable medical mask, surgical mask, dust mask,FFP2, FFP3, KN95 mask, PM2.5 mask, nano mask, fresh air mask, kids mask ;antigen self-test kit. Medical protective clothing, civil protective clothing, disposable isolation clothing, virus protective clothing, brush hand suit, clean clothing, hand washing clothes, operation clothes, surgical clothes, surgical clothes, doctor nurse clothes, sterile caps, surgical caps, shoe covers, medical gloves, medical rubber gloves, sterile film hand covers, sterile drug giving fingersets, inspection gloves, latex gloves, PE gloves, PVC gloves, Nitrile gloves, Isolation protection: medical
isolation shoes, medical isolation shoes,medical isolation goggles, disposable protective boot isolation cabin, etc; Anti stick cloth, melt spray cloth, nose beam strip, elastic
ear belt, protective clothing fabric, kn95 mask machine, disposable flat mask production line. The company has established a high level of quality management system to ensure the safety and quality of products! Business cooperation mode: brand global investment agent, ODM, OEM.
ear belt, protective clothing fabric, kn95 mask machine, disposable flat mask production line. The company has established a high level of quality management system to ensure the safety and quality of products! Business cooperation mode: brand global investment agent, ODM, OEM.
Certificatie

Exhibition Cooperation
FAQ
Q: When can I test myself?
A: You can always test yourself whether vou have symptoms or not. Please note that the test result is a snapshot that is valid for this point in time. Tests should therefore be repeated according to the regulations of the responsible authorities.
Q: What should I pay attention to in order to obtain the most exact test result possible?
A: Always follow the instructions for use exactly. Perform the test immediately after collecting the sample. Dispense the drops from the test tube only into the designated well of the test cassette. Dispense 2-3 drops from the sample tube. Too many or too few drops can lead to an incorrect or invalid test result
Q: The test strip is discolored. What is the reason or what am I doing wrong?
A: The reason for a clearly visible discoloration of the test strip is that too large a quantity of drops has been dispensed from the sample tube into the test devices well. The indicator strip can only hold a limited amount of liquid. If the control line does not appearor the test strip is discolored. please repeat the test with a new test kit according to the instructions for use
Q: What should I do if I took the test but didn't see a control line?
A: In this case, the test result is to be considered invalid. Please repeat the test with a new test kit according to the instructions for use
Q: I am unsure of the interpretation of the results. What should I do?
A: If you cannot clearly determine the result of the test, contact the nearest medical facility applying the regulations of your local authority.
Q: My result is positive. What should I do?
A: If a red strip appears both on the control line (C-line) and the test line (T-line) of the cassette, your result is positive and you should immediately contact the medical facility in accordance with the requirements of your local authorities. Your test result may be checked and the next steps will be explained to you
Q: My result is negative. What should I do?
A: If a red strip appears only at the control line (C-line) of the cassette, this may mean that you are negative or that the viral load is too low to be recognized by the test. If you experience symptoms such as headaches, migraines, fever, loss of sense of smell and taste, contact the nearest medical facility applying the regulations of your local authority. In addition, you can repeat the test with a new test kit.
Q: Can this test cassette be reused or used by multiple people?
A: This test cassette is for one-time use and cannot be reused or used by multiple people
A: You can always test yourself whether vou have symptoms or not. Please note that the test result is a snapshot that is valid for this point in time. Tests should therefore be repeated according to the regulations of the responsible authorities.
Q: What should I pay attention to in order to obtain the most exact test result possible?
A: Always follow the instructions for use exactly. Perform the test immediately after collecting the sample. Dispense the drops from the test tube only into the designated well of the test cassette. Dispense 2-3 drops from the sample tube. Too many or too few drops can lead to an incorrect or invalid test result
Q: The test strip is discolored. What is the reason or what am I doing wrong?
A: The reason for a clearly visible discoloration of the test strip is that too large a quantity of drops has been dispensed from the sample tube into the test devices well. The indicator strip can only hold a limited amount of liquid. If the control line does not appearor the test strip is discolored. please repeat the test with a new test kit according to the instructions for use
Q: What should I do if I took the test but didn't see a control line?
A: In this case, the test result is to be considered invalid. Please repeat the test with a new test kit according to the instructions for use
Q: I am unsure of the interpretation of the results. What should I do?
A: If you cannot clearly determine the result of the test, contact the nearest medical facility applying the regulations of your local authority.
Q: My result is positive. What should I do?
A: If a red strip appears both on the control line (C-line) and the test line (T-line) of the cassette, your result is positive and you should immediately contact the medical facility in accordance with the requirements of your local authorities. Your test result may be checked and the next steps will be explained to you
Q: My result is negative. What should I do?
A: If a red strip appears only at the control line (C-line) of the cassette, this may mean that you are negative or that the viral load is too low to be recognized by the test. If you experience symptoms such as headaches, migraines, fever, loss of sense of smell and taste, contact the nearest medical facility applying the regulations of your local authority. In addition, you can repeat the test with a new test kit.
Q: Can this test cassette be reused or used by multiple people?
A: This test cassette is for one-time use and cannot be reused or used by multiple people
We Recommend
New Arrivals
New products from manufacturers at wholesale prices