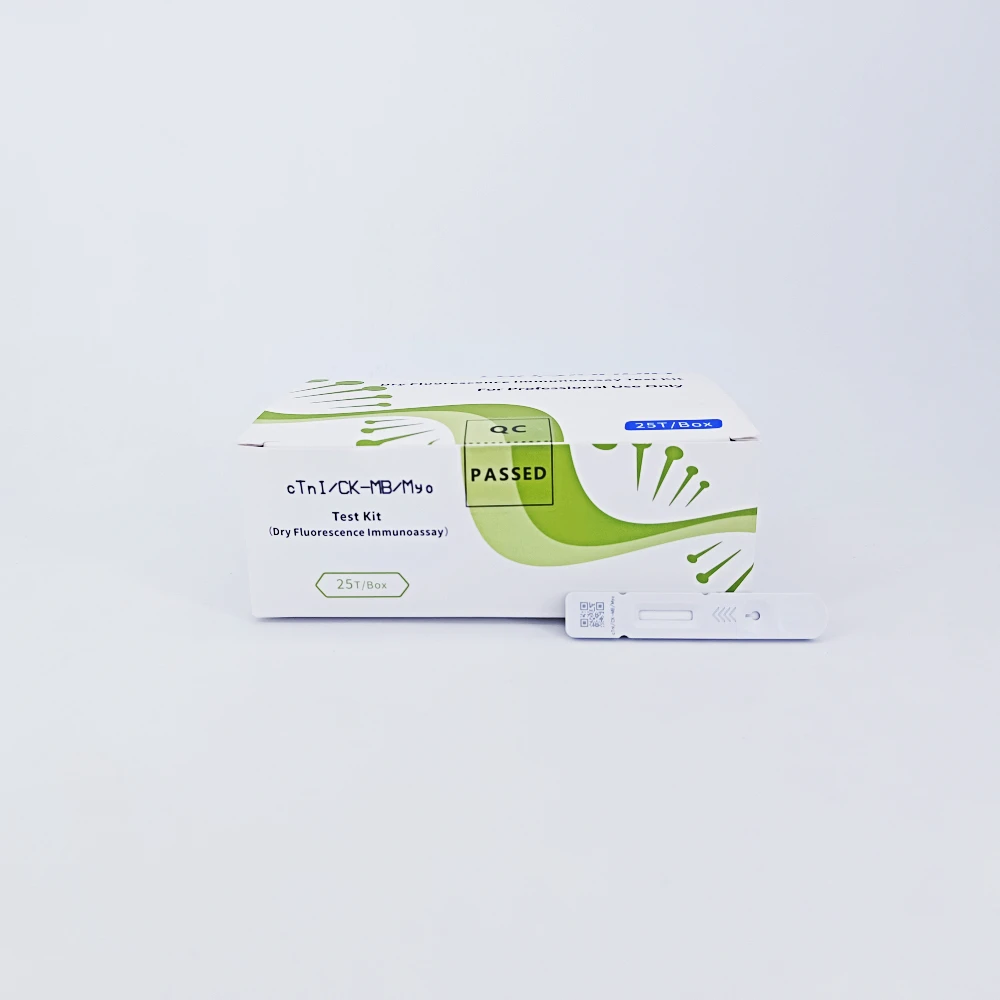
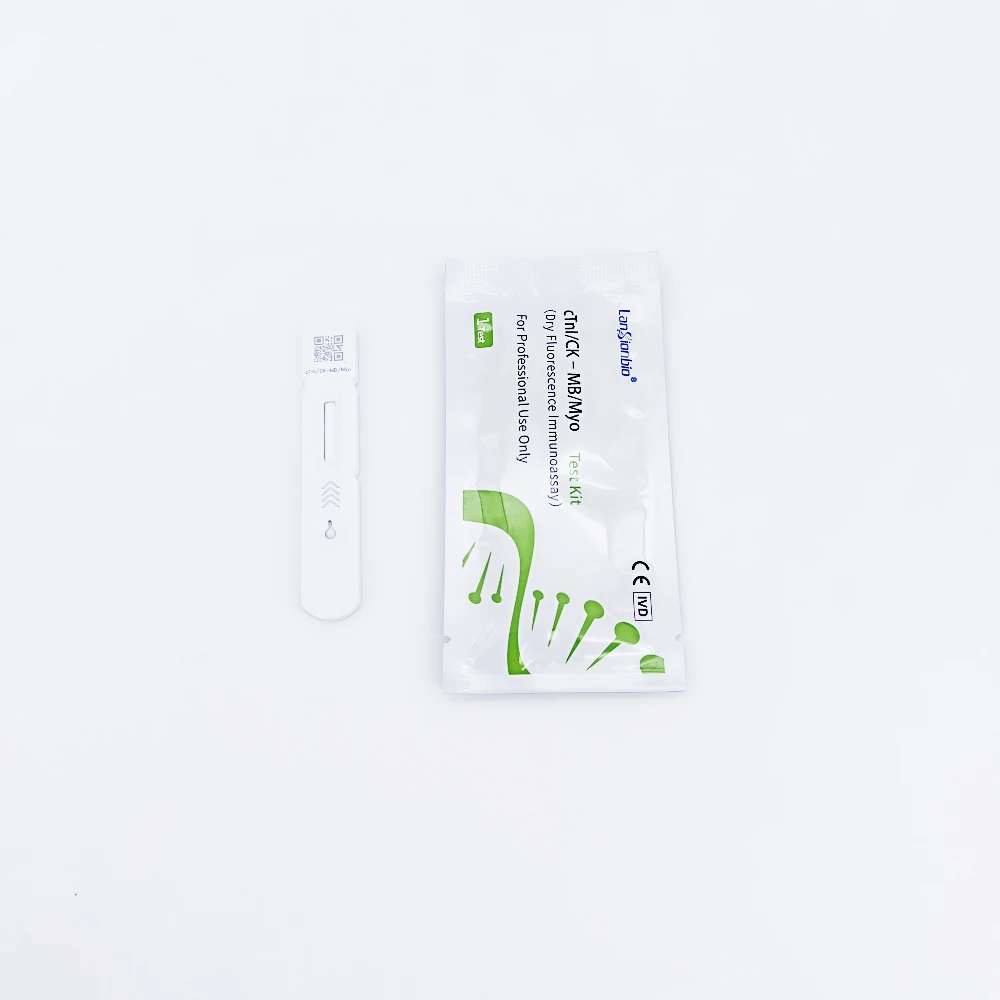
Lansionbio cTnI/CK MB/Myo Rapid Test Kit Diagnostic Combo Test Cassette with CE ISO Certification
- Category: Clinical Analytical Instruments >>>
- Supplier: Lansion Biotechnology Co. Ltd.
Share on (1600515754414):
Product Overview
Description
Product Description

(Dry Fluorescence Immunoassay)
cTnI /CK-MB/Myo Test Kit is intended for in-vitro quantitative measurement of cTnI /CK-MB/Myo in human serum.
This test is used for the detection for clinical assistance in diagnosis.
cTnI is used as an aid in the diagnosis of myocardial injury such as Acute Myocardial Infarction (AMI),Unstable Angina, Acute Myocarditis and Acute Coronary Syndrome (ACS).
CK-MB is an important indicator for the diagnosis of acute myocardial infarction (AMI).
Myo can be used as the most sensitive index in the early diagnosis of acute myocardial infarction (AMI).

cTnI /CK-MB/Myo Test Kit : | |
Place of Origin | China |
Jiangsu | |
Brand Name | Lansionbio |
Model Number | IVD poct reagent |
Power Source | Manual |
Warranty | 1 Year |
After-sale Service | Online technical support |
Material | Plastic |
Shelf Life | 1.5 years |
Quality Certification | CE, ISO13485 |
Instrument classification | Class II |
Test item | cTnI /CK-MB/Myo |
Type | Immunoassay System |
Brand Name | Lansionbio |
Methodology | TRFIA |
Test Principle | Antigen-antibody reaction |
Reaction time | 10 minutes |
Storage Temperature | 4-30 °C |
Application | Hospital, Clinic, Laboratory |
Sample Type | Serum |
Shelf Life | 18 Months |


Diabetes: | HbA1c |
Inflammation: | CRP PCT SAA SAA/CRP PCT/CRP IL-6 |
Cardiac: | CK-MB cTnI Myo cTnI/CK-MB/Myo NT-proBNP D-Dimer H-FABP |
Hormone: | TT3 TT4 TSH 25-OH-VD β-HCG LH FSH PRL AMH |
Gastric Function: | PGI/PGII |
Anemia: | Ferritin |
Tumour: | PSA |


As a high-tech biotechnology company, Lansion Biotechnology Co., Ltd.(Lansionbio) is speialized in research &
development,production, sell, services of in vitro diagnostic reagents and devices. And its members of core R&D team all come from well-come research institutions and bio-pharmaceutical companies in and abroad. With its advanced technology concept, Lansionbio provides equioment, reagents and services to world-wild clients according to their needs.
With its international leading technology, Lansionbio has obtained ISO13485 certificate and built a R&D center covering 7500 square meters, which also has a 2400 square meters aseptic purification workshop meeting GMP standard. Presently Lansionbio has 2 subsidiaries: Suzhou Lansion Biotechnology Co., Ltd and Jiangsu Huao Lansion Biotechnology Co.,Ltd, providing products including IVD devices and reagents.
Lansionbio has been always upholding the innovation, committing to breakthrough, insisting the R&D work from beginning. Now Lansionbio has already built up 3 world-leading platforms: microfluidics, immunochromatography, molecular diagnosis. Lansionbio has won great trusts from numerous customers by its world-leading quality standards.
Lansionbio takes "Strive for excellence, care every life and health" as purpose, focuses on innovation and quality and devotes to improve the human health by providing high-quality, reliable and affordable POCT produts and services.
development,production, sell, services of in vitro diagnostic reagents and devices. And its members of core R&D team all come from well-come research institutions and bio-pharmaceutical companies in and abroad. With its advanced technology concept, Lansionbio provides equioment, reagents and services to world-wild clients according to their needs.
With its international leading technology, Lansionbio has obtained ISO13485 certificate and built a R&D center covering 7500 square meters, which also has a 2400 square meters aseptic purification workshop meeting GMP standard. Presently Lansionbio has 2 subsidiaries: Suzhou Lansion Biotechnology Co., Ltd and Jiangsu Huao Lansion Biotechnology Co.,Ltd, providing products including IVD devices and reagents.
Lansionbio has been always upholding the innovation, committing to breakthrough, insisting the R&D work from beginning. Now Lansionbio has already built up 3 world-leading platforms: microfluidics, immunochromatography, molecular diagnosis. Lansionbio has won great trusts from numerous customers by its world-leading quality standards.
Lansionbio takes "Strive for excellence, care every life and health" as purpose, focuses on innovation and quality and devotes to improve the human health by providing high-quality, reliable and affordable POCT produts and services.











Add : No.2, Qiande Road, Science Park, Jiangning District, Nanjing, Jiangsu Province, P. R. CHINA
Tel : 86-25-58577600
Fax : 86-25-58758600
Website : //en.lansionbio.com
Fax : 86-25-58758600
Website : //en.lansionbio.com
We Recommend
New Arrivals
New products from manufacturers at wholesale prices