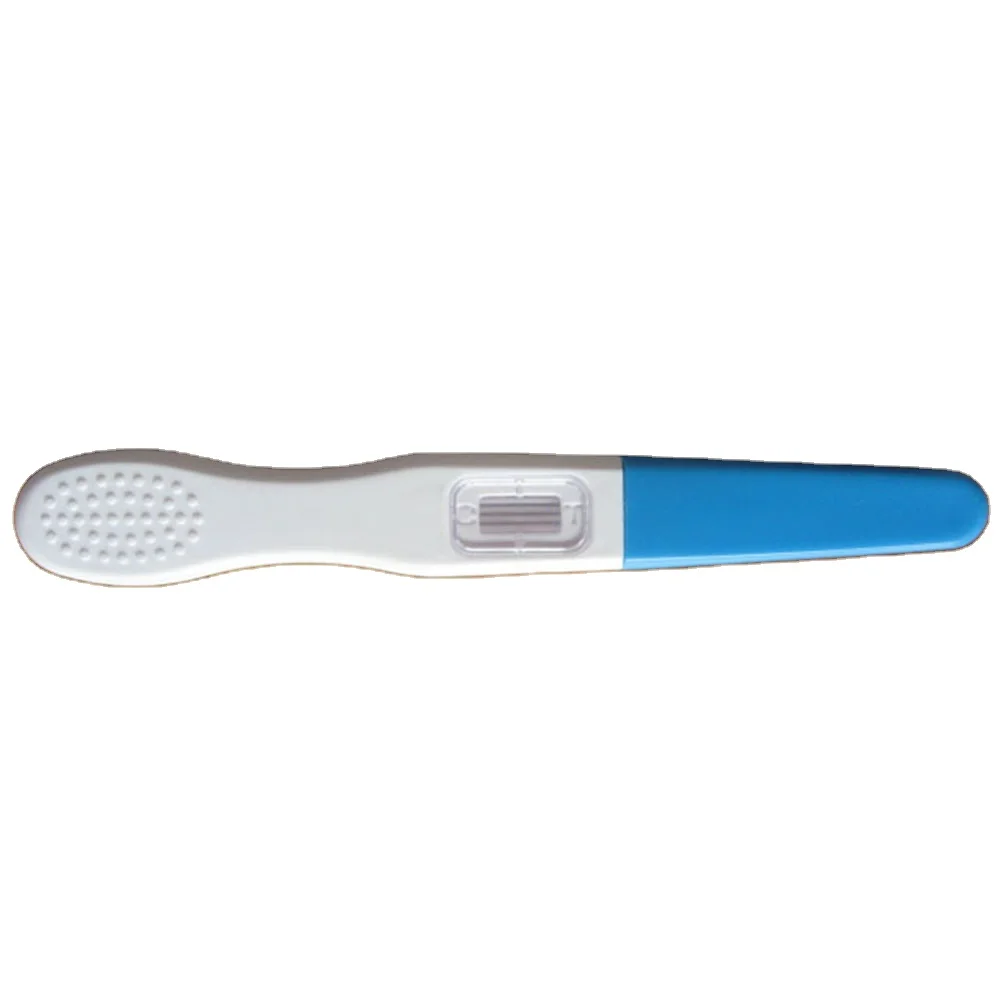
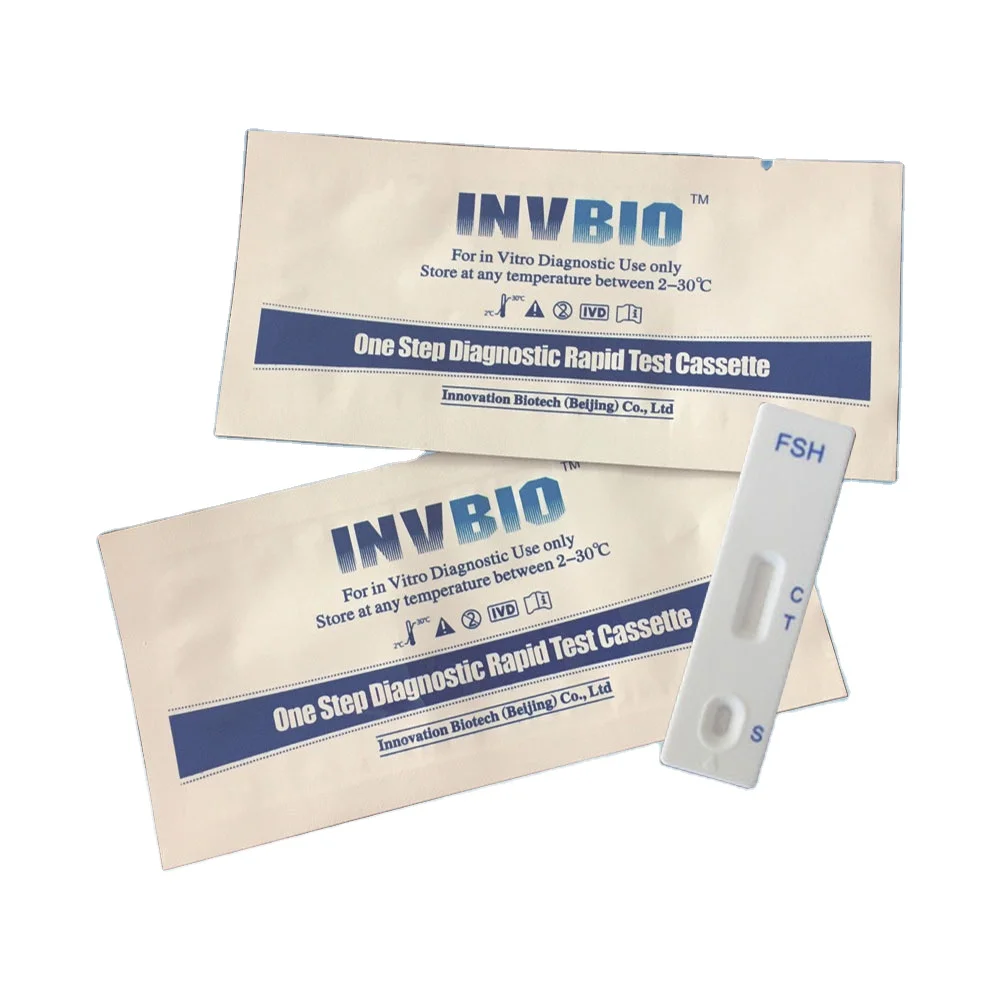
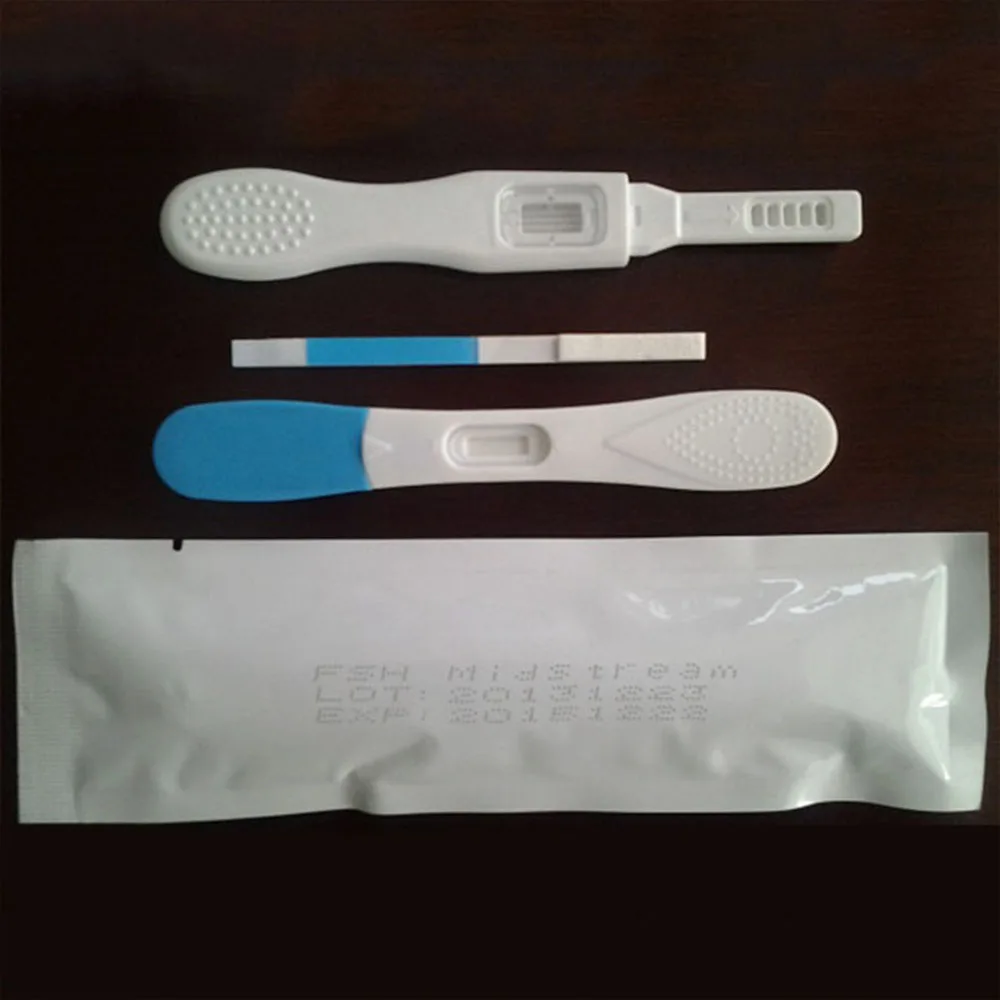
ce test lh fsh hcg rapid diagnostic kit
- Category: >>>
- Supplier: Beijing Icon Technology Co. Ltd.
Share on (1600549719886):
Product Overview
Description
Specification
item | value |
Place of Origin | China |
Brand Name | INVBIO |
Model Number | INV-133 |
Type | Pathological Analysis Equipme |
Specimen | Urine |
Format | midstream |
Sensitivity | 25mIU/ml, 20mIU |
Accuracy | >99% |
Package | Individual Foil Pouch |
Storage | Room Temperature 4-30 |
Reading time | Within 5 minutes |
Method | Colloidal Gold Rapid Test |
Product Description
Packing & Delivery


Company Profile



BEIJING ICON TECHNOLOGY CO., LTD is a biotechnology company specializing in research, development and manufacturing of advanced medical in-vitro diagnostic (IVD) rapid test kits, medical and laboratory disposal products. We provide one step medical diagnostic rapid test kit, Product include Fertility Tests, Infectious diseases, Tumor markers, DOA drug of abuse test, Drug test cup, Urinalysis reagent strip, ELISA kit, Digital alcohol tester, urine analyzer, et.
OEM packaging is available, drug of abuse test, troponin I test, alcohol screening saliva test strips, urine strips, elisa kits
Our focus is to expand our markets internationally by forming strategic alliances and entering into partnerships with distributors worldwide. We have established an international reputation for excellence in the manufacturing of quality medical and laboratory products. In addition, we have obtained approval licenses ISO13485, FSC certificate, and most of our products get CE mark Our objective is the utmost satisfaction of our clients all around the world by supplying our quality and economical products
OEM packaging is available, drug of abuse test, troponin I test, alcohol screening saliva test strips, urine strips, elisa kits
Our focus is to expand our markets internationally by forming strategic alliances and entering into partnerships with distributors worldwide. We have established an international reputation for excellence in the manufacturing of quality medical and laboratory products. In addition, we have obtained approval licenses ISO13485, FSC certificate, and most of our products get CE mark Our objective is the utmost satisfaction of our clients all around the world by supplying our quality and economical products

ExpoMedical exhibition in Argentina
Date: from September 26, 2018 to September 28, 2018
Add.: Centro Costa Salguero, Buenos Aires, Argentina
Our booth No: C12 in Hall 4
MEDICA in Germany
Date: from November 12th to November 15th
Add.: Messe Düsseldorf
Out booth No: will update in June
Date: from September 26, 2018 to September 28, 2018
Add.: Centro Costa Salguero, Buenos Aires, Argentina
Our booth No: C12 in Hall 4
MEDICA in Germany
Date: from November 12th to November 15th
Add.: Messe Düsseldorf
Out booth No: will update in June
FAQ
1. What certificates do you have?
We have a professional CE certificate and Declaration of Conformity. Our antigen test have passed the German PEI testing and got Bfarm number(the white list of the German food and Drug Administration) and antigen nasal test has registered on EU common list. There is no problem selling them in Europe. The CE announcement number is for self-test products, and we only have a professional test certificate.
We have a professional CE certificate and Declaration of Conformity. Our antigen test have passed the German PEI testing and got Bfarm number(the white list of the German food and Drug Administration) and antigen nasal test has registered on EU common list. There is no problem selling them in Europe. The CE announcement number is for self-test products, and we only have a professional test certificate.
2. Do you have CE certificate of self testing?
Our JOUNT-VENTRUED COMPANY have CE certificate of self testing.
3. How can we guarantee quality?
Always a pre-production sample before mass production;
Always final Inspection before shipment.
Always a pre-production sample before mass production;
Always final Inspection before shipment.
4. What about the payment methods?
T/T, Trade Assurance, Paypal, Western Union
T/T, Trade Assurance, Paypal, Western Union
5. Can I print our logo on the package?
Yes, we can do custom printing on pouches and boxes.
Yes, we can do custom printing on pouches and boxes.
6. Can you provide OEM?
Yes, OEM is available. All of our products can be customized.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices