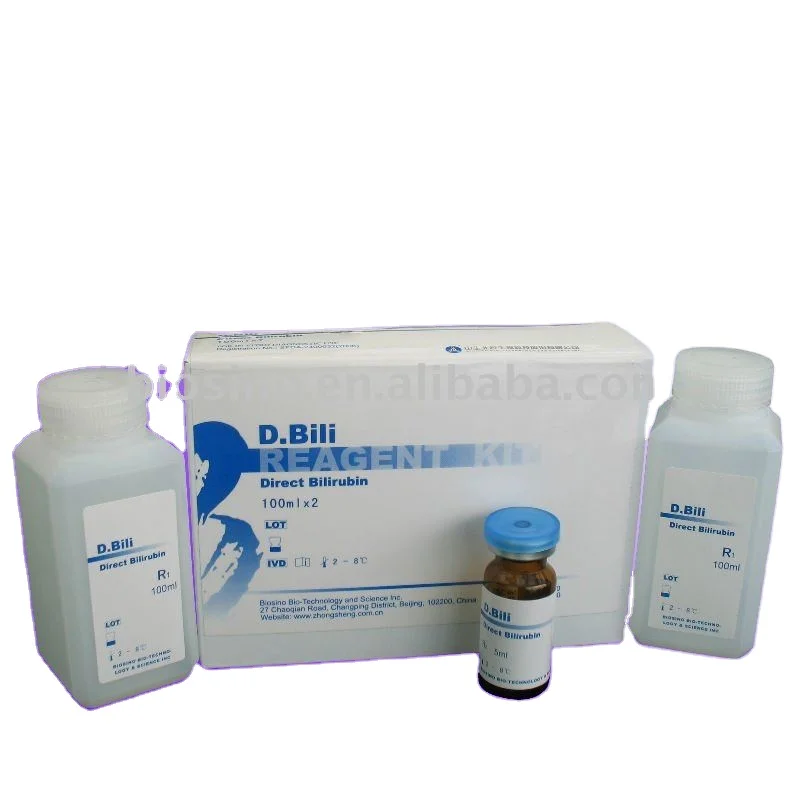
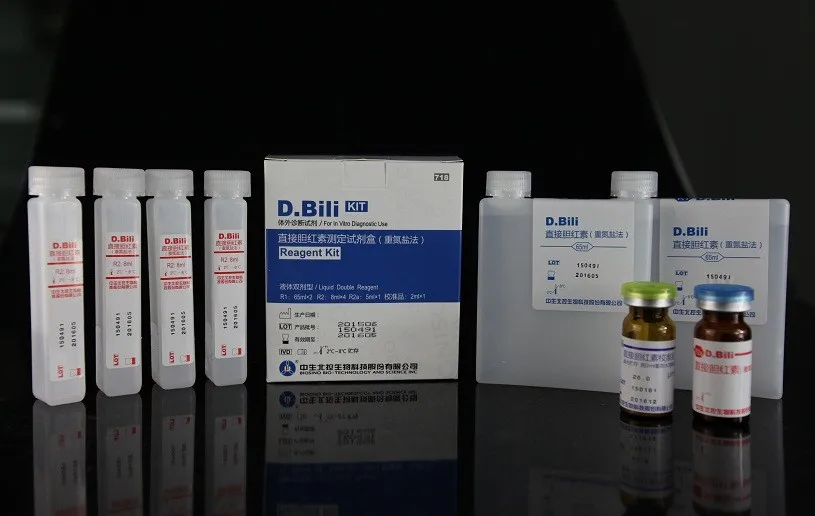
Direct Bilirubin D.Bili diagnostic reagents, Direct Bilirubin D.Bili Activity Assay Kit
- Category: >>>
- Supplier: BioSino Bio-Technology And Science Inc.
Share on (1600623192208):
Product Overview
Description
Product Description
This product is used for the quantitative determination of direct bilirubin in human serum on clinical chemistry analyzers.
Bilirubin is a complex linear molecule composed of four pyrrole rings. It is a metabolite of heme in the body. According to the different forms in the body, it is divided into two categories: indirect bilirubin and direct bilirubin.
In clinical, direct bilirubin index is mainly used for the diagnosis of jaundice and the judgment of the type of jaundice. Since bilirubin metabolism is closely related to liver function, in practical applications, direct bilirubin detection is often combined with total bilirubin detection as an important routine liver function index to judge liver function status and jaundice.
The commonly used methods for clinical detection of direct bilirubin indexes include diazonium salt method, vanadate oxidation method and bilirubin oxidase method. This product is based on the vanadate oxidation method, which has the advantages of high sensitivity, good repeatability and strong anti-interference ability.
Specification
Item | value |
Type | Biochemical Analysis System |
Brand Name | BIOSINO |
Model Number | 100020130 100020135 |
Test Function | Liver Test |
Product name | Direct Bilirubin D.Bili Reagent |
Method | Vanadate Oxidation Method |
Contents | R1: 60ml×4, R2: 15ml×4, calibrator: 0.5ml×1 R1: 80ml×2, R2: 20ml×2, calibrator: 0.5ml×2 R1: 48ml×3, R2: 36ml×1, calibrator: 0.5ml×1 |
Format | Double Liquid |
ROCHE HITACHI 7180/7170/7060/7080/7600P OLYMPUS AU 400/640/2700/5400 BECKMAN DXC-800、BECKMAN COULTER CX5 ABBOTT ARCHITECT c4000/c8000/c16000 TOSHIBA 40FR/120FR | |
Valid Time | 2 Years |
After-sale Service | 24 hours Online technical support |
Packing & Delivery


transport by air with icebag, cold-chain transport
Company Profile



BioSino Biotechnology & Science Inc. is one of the largest manufacturers of clinical diagnostic reagents in China, with more than 20 years of history. Our company was founded by IBP, one of the top research institutes in China. We are the first diagnostic company of China Listed in Hong Kong Stock Exchange. We are ISO9001/13485 certified.
We are the first company developing diagnostic enzyme reagent kit and liquid double reagents for Hitachi Analyzers in China.
We are one of the biggest GMP standard factories (11000 sq.m.) in China and have automatic production facilities, with a daily capacity of 6000 kits of reagents.
We are the first Chinese diagnostic company operated with ERP system.
We are the first Chinese company establishing reference material Lab and finishing the systematization of reagents, equipment and reference materials.
Biosino has 90 series and more than 200 kinds of IVD products, including biochemistry diagnostic reagents, immunology diagnostic reagents and rapid tests. Lots of our products have got CE mark. BioSino has developed over 600 distributors and retailers over the country, occupying 75% of domestic market. Moreover, we have exported our products to over 40 countries and regions, such as Southeast Asia, the Middle East, North Africa and South America.
FAQ
1. who are we?
We are based in Beijing, China, start from 1988,sell to Domestic Market(50.00%),Mid East(12.00%),Southeast Asia(8.00%),Western Europe(5.00%),Eastern Europe(4.00%),South Asia(3.00%),Southern Europe(3.00%),South America(3.00%),Eastern Asia(3.00%),Northern Europe(2.00%),Central America(2.00%),Oceania(2.00%),North America(2.00%),Africa(1.00%). There are total about 501-1000 people in our office.
2. how can we guarantee quality?
Always a pre-production sample before mass production;
Always final Inspection before shipment;
3.what can you buy from us?
Clinical chemistry reagents,In Vitro diagnostic reagents,clinical chemistry analyzers,rapid test kit,Elisa Kit
4. why should you buy from us not from other suppliers?
BioSino is certified with ISO9001 and ISO13485, and the manufacturer is built according to GMP standards. Over 500 distributors and retailers covering 75% of the users in the industry,registered the clinical chemistry reagents in CMDCAS of Canada
5. what services can we provide?
Accepted Delivery Terms: FOB,CFR,CIF,EXW;
Accepted Payment Currency:USD,EUR,RMB;
Accepted Payment Type: T/T;
Language Spoken:English,Chinese
We are based in Beijing, China, start from 1988,sell to Domestic Market(50.00%),Mid East(12.00%),Southeast Asia(8.00%),Western Europe(5.00%),Eastern Europe(4.00%),South Asia(3.00%),Southern Europe(3.00%),South America(3.00%),Eastern Asia(3.00%),Northern Europe(2.00%),Central America(2.00%),Oceania(2.00%),North America(2.00%),Africa(1.00%). There are total about 501-1000 people in our office.
2. how can we guarantee quality?
Always a pre-production sample before mass production;
Always final Inspection before shipment;
3.what can you buy from us?
Clinical chemistry reagents,In Vitro diagnostic reagents,clinical chemistry analyzers,rapid test kit,Elisa Kit
4. why should you buy from us not from other suppliers?
BioSino is certified with ISO9001 and ISO13485, and the manufacturer is built according to GMP standards. Over 500 distributors and retailers covering 75% of the users in the industry,registered the clinical chemistry reagents in CMDCAS of Canada
5. what services can we provide?
Accepted Delivery Terms: FOB,CFR,CIF,EXW;
Accepted Payment Currency:USD,EUR,RMB;
Accepted Payment Type: T/T;
Language Spoken:English,Chinese
We Recommend
New Arrivals
New products from manufacturers at wholesale prices