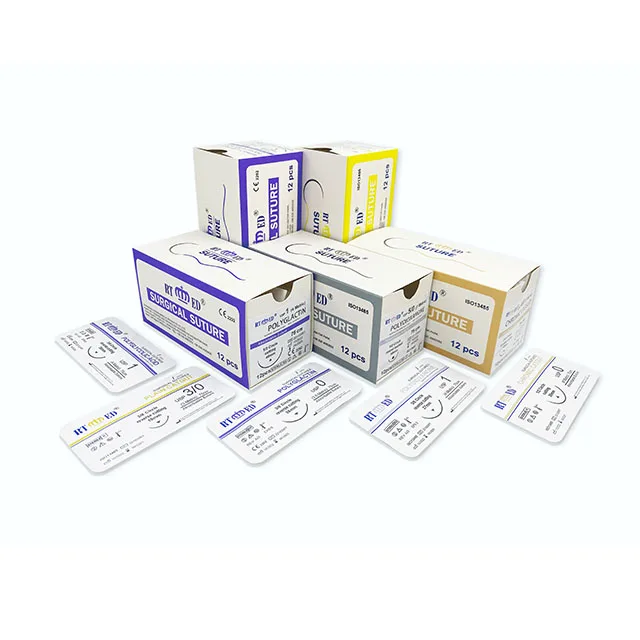
Wego CE/ISO Certification 100%polyglycolic acid Absorbable Surgical Medical Suture With Needle PGA PDO PGCL
- Category: >>>
- Supplier: Shandong Weigao Group Medical Polymer Co. Ltd.
Share on (1600948717624):
Product Overview
Description
Poster

Products Description



Specification
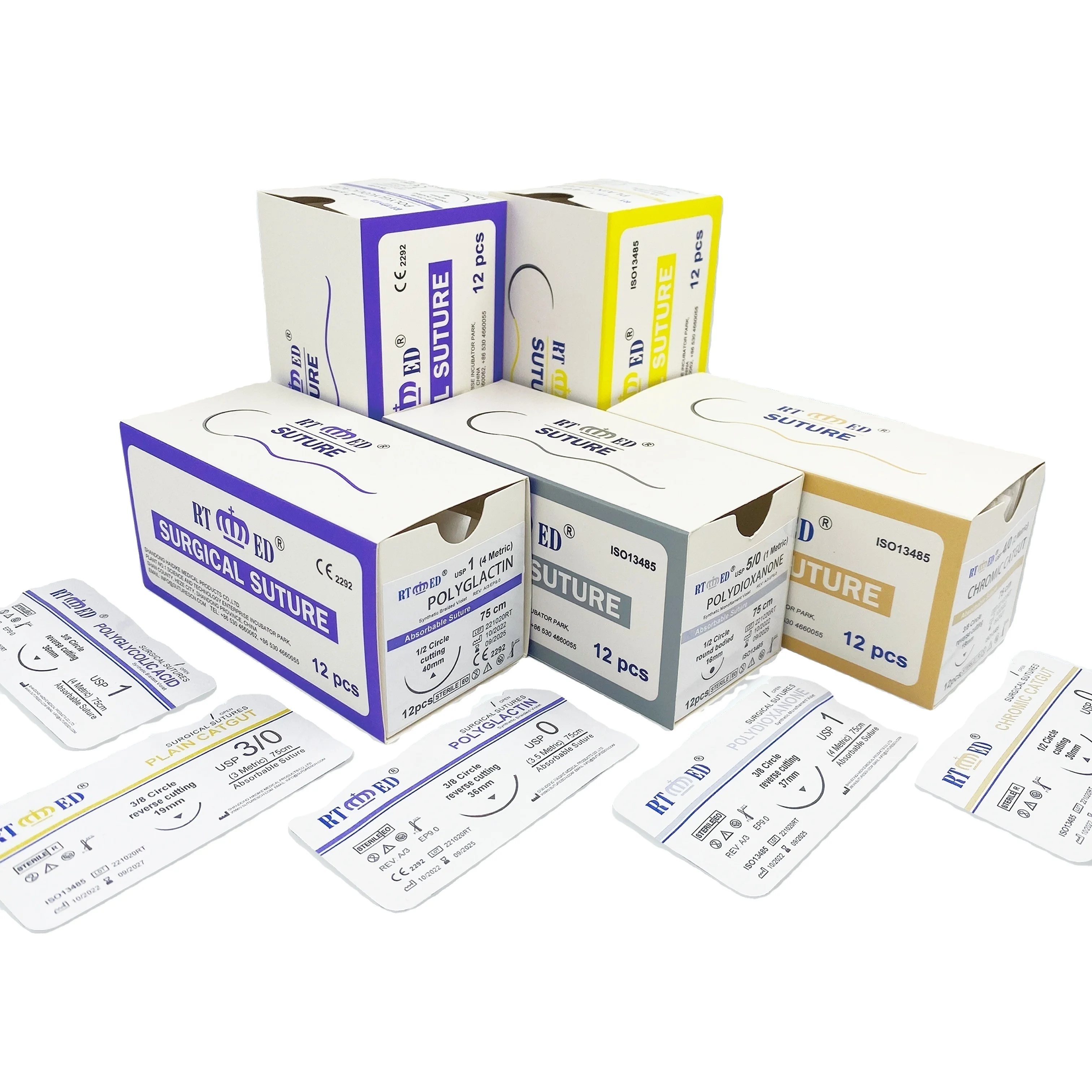
Product Description | WEGOSUTURES already obtain CE mark and USA FDA 510K. WEGOSUTURES conformity with the USP and EP standard. suture needle Products including Braided PGA, Braided PGLA, Braided Rapid PGA, Monofilament PDO, Silk, Nylon, Polyester and Polypropylene. WEGOSUTURE if soft and easy for safe knot, absorbed by the tissues with hydrolysis after implant, safe and without histological reactions. Sterile by EO conformity with ISO11137. Mainly markets are US and European markets. Over 6 years exported experience and obtained good reputation from customers. our sutures products covers the full range of sutures sizes: from USP 7# to USP 12/0 including varies type of needles or non-needle products. Sutures type including absorbable and non absorbable. Special sutures are available inlcuding Cardiovascular sutures, Ophthalmic sutures and etc. |
WEGO-PGA Features | Structure Multifilament, braided Chemical composition 100%polyglycolic acid Coating Polycaprolactone + Calcium Stearate Color Violet or Undyed Sizes USP 2-USP 8/0 metric 5 - metric 0.4 Knot tensile strength Retention 14 days post implantation 60-70% 18 days post implantation 50% 21 days post implantation 40% Mass absorption Degradation by hydrolysis within 60-90 days Indications General surgery, Gastroenterology, Urology, Gynaecology, Ophthalmic, Plastic and Paedriatic surgery Sterilization Ethylene oxide |
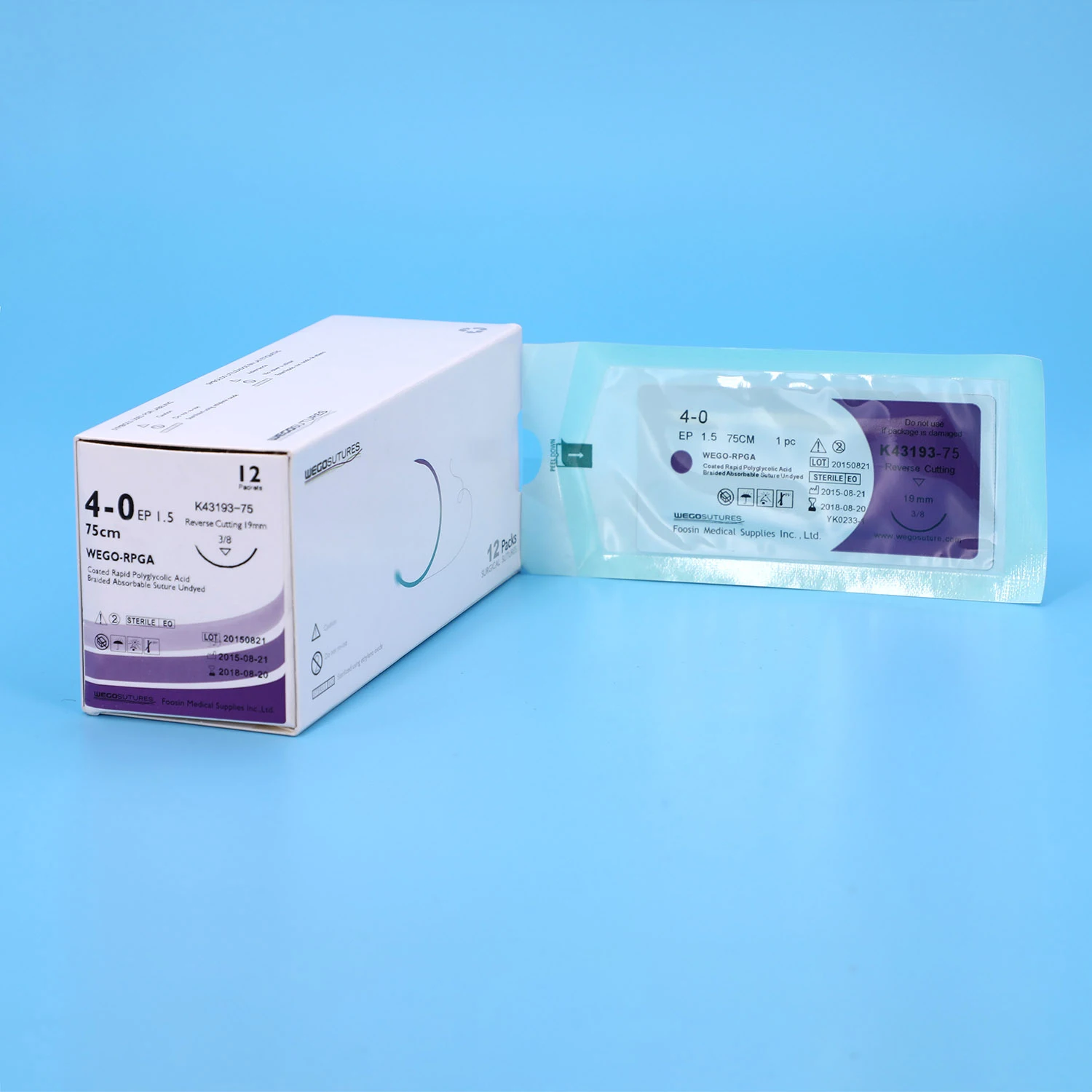
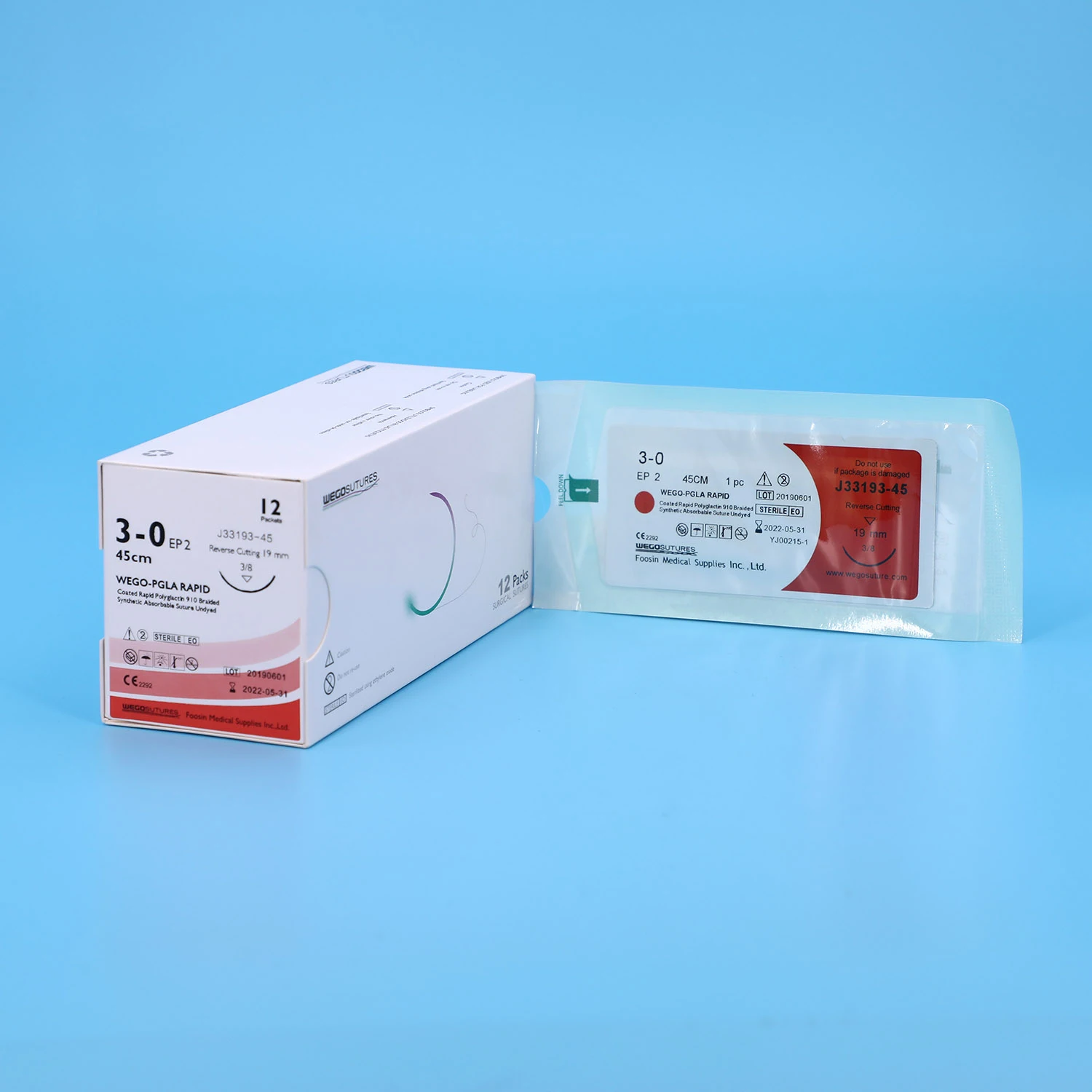
WEGO-PGA Rapid Features | WEGO-PGA Rapid sutures are synthetic, absorbable, sterile surgical sutures composed of Polyglycolic Acid (PGA). The characteristic rapid loss of strength is achieved by use of a polymer material with a lower molecular weight than regular WEGO-PGA suture. |
WEGO-NYLON Features | Structure Monofilament Chemical composition Polyamide 6 and 6.6 Color Blue, Undyed and Black Sizes USP 2- USP 10/0 metric 2 - metric 0.1 Knot tensile strength Retention 15-20% per year Indications Skin and Plastic Surgery, Micro-vascular surgery, Ophthalmology, Orthopaedics Contraindications Not known. Sterilization Ethylene oxide |
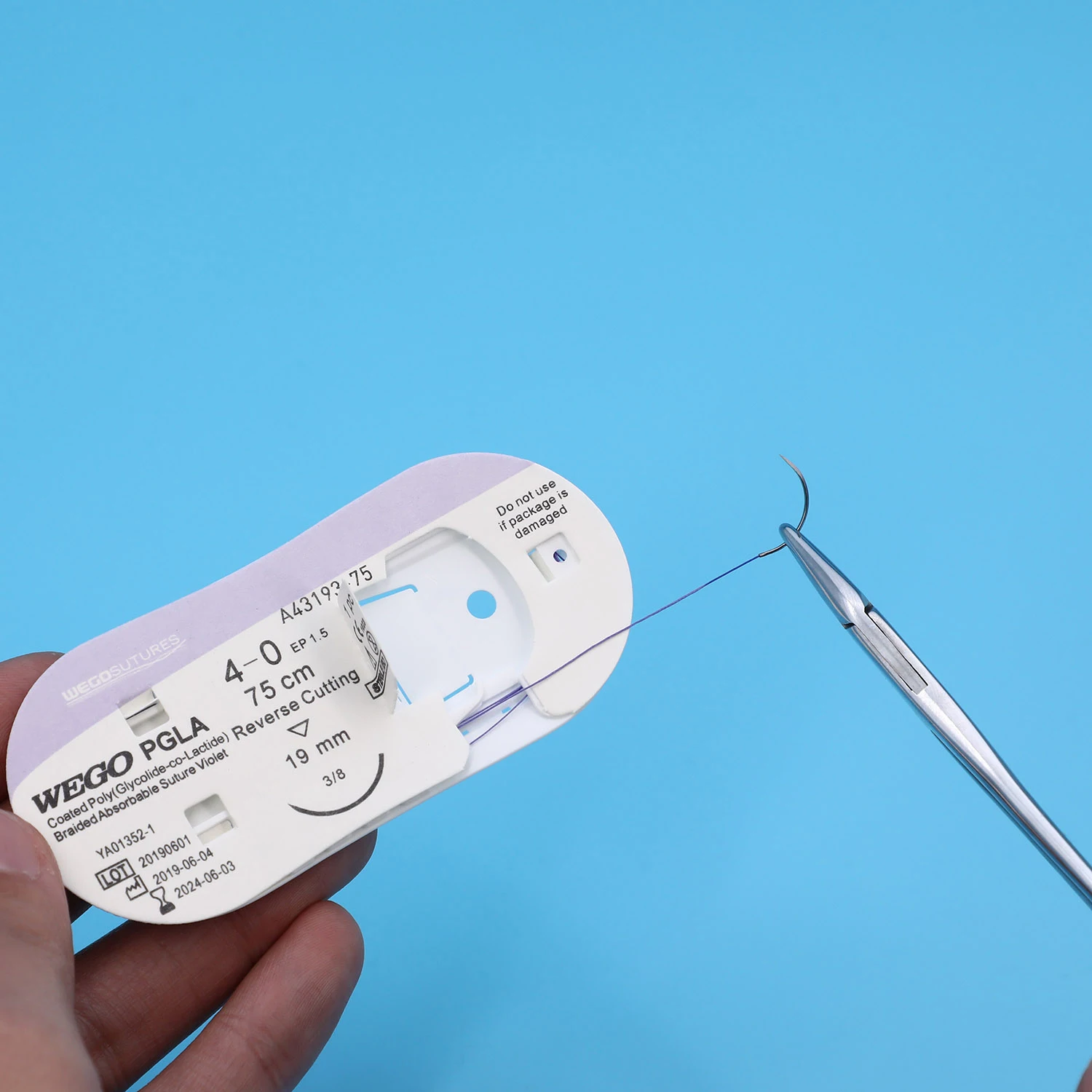
WEGO-PGLA Features | Structure:Monofilament, braided. Chemical composition:Poly(glycolide-co-lactide)(90/10). Coating:Poly(glycolide-co-lactide)(30/70)+Calcium Stearate. Color:Violet ( D&C violet No. 2). Sizes:USP 2- USP 7/0 metric 5 - metric 0.5. Knot tensile strength Retention:14days post implantation 75%./21days post implantation 50%. Mass absorption:Degradation by hydrolysis within 56~70 days. Indications:Subcutaneous and intracutaneous closure, Gynaecology, Odontology, Paediatric surgery, Ophthalmology, Ligatures. Sterilization:Ethylene oxide. |
WEGO-PGLA Rapid Features | WEGO-PGLA Rapid sutures are synthetic, absorbable, braided, sterile surgical sutures composed of a copolymer made from 90% glycolide and 10% L-lactide. The characteristic rapid loss of strength is achieved by use of a polymer material with a lower molecular weight than regular WEGO-PGLA (Polyglactin 910) suture. |
WEGO-PDO Features | Structure: Monofilament Chemical composition:Polydioxanone Color:Violet(D&C No.2) or Undyed Sizes :USP 2- USP 7/0 (metric 5 – metric 0.5) Retention:14 days post implantation 75% 28 days post implantation 65% 42 days post implantation 50% Mass absorption:Degradation by hydrolysis within 180 – 220 days Indications:General soft tissue approximation, including use in paediatric cardiovascular tissue where growth is expected to occur and in ophthalmic surgery. These sutures are particularly useful where the combination of an absorbable suture and extended wound support (up to six weeks) is desirable Contraindications:These sutures, being absorbable, should not be used where prolonged (beyond 6 weeks) approximation of tissues under stress is required and is not to be used in conjunction with prosthetic devices, for example, heart valves or synthetic grafts. WEGO-PDO suture is not indicated in adult cardiovascular tissue, microsurgery and neural tissue. |
WEGO-PGCL Features | Structure :Monofilament Chemical Composition:Poly(glycolideco-caprolactone) (PGA-PCL) Color:Undyed or Violet(D&C no.2) Sizes :USP 2- USP 6/0 (metric 5 – metric 0.7) Strength Retention Profile:7 days post implantation 65% 14 days post implantation 40% Absorption Profile:Degradation by hydrolysis within90-110days Indications:General soft tissue approximation and/or ligation where an absorbable material is indicated. |




Company Profile
FAQ
1.Q: Is your company a factory or trading company?
A: Our company is a professional manufactory .WEGO is selected as one of China's Top 500 Enterprises (Ranked 487th) and China's Top 500 Manufacturing Enterprises (Ranked 257th).
2.Q:Can I have a sample first ?
A:Sure!Sample are available and free with certain QTY,if not,we'll charge accordingly.But the express fee need to paid by your esteemed company.
3.What is delivery time?
A: Our company is a professional manufactory .WEGO is selected as one of China's Top 500 Enterprises (Ranked 487th) and China's Top 500 Manufacturing Enterprises (Ranked 257th).
2.Q:Can I have a sample first ?
A:Sure!Sample are available and free with certain QTY,if not,we'll charge accordingly.But the express fee need to paid by your esteemed company.
3.What is delivery time?
A:Usually 30-45 days.
4.Q:What is terms of payment?
A: 1) 30% T/T in advance as for deposit, balance before delivery.
2)100% L/C at sign
5.Q: Can I visit your factory?
A: Sure, warmly welcome your visit to WEGO. Please try to contact our sales representative first by email or phone. We will make the appointment and the most reasonable planning for your transportation.
6.Q: What's the shipping way?
A: Regarding the small quantity order, we can send them by Fedex,DHL, express and the large quantity goods are shipped by ocean shipping according to client's demands.
4.Q:What is terms of payment?
A: 1) 30% T/T in advance as for deposit, balance before delivery.
2)100% L/C at sign
5.Q: Can I visit your factory?
A: Sure, warmly welcome your visit to WEGO. Please try to contact our sales representative first by email or phone. We will make the appointment and the most reasonable planning for your transportation.
6.Q: What's the shipping way?
A: Regarding the small quantity order, we can send them by Fedex,DHL, express and the large quantity goods are shipped by ocean shipping according to client's demands.
7.Q: Do you provide OEM/ODM Service ?
A:Yes,please Send your requirements in details,we provide custom-make service to meet different needs of global clients with different budgets.
A:Yes,please Send your requirements in details,we provide custom-make service to meet different needs of global clients with different budgets.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices