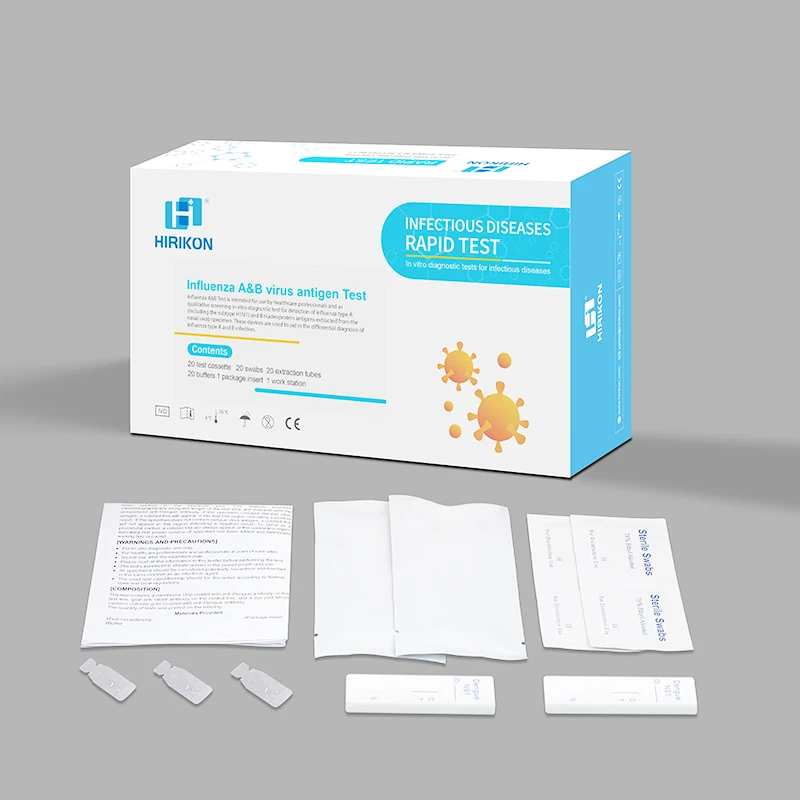
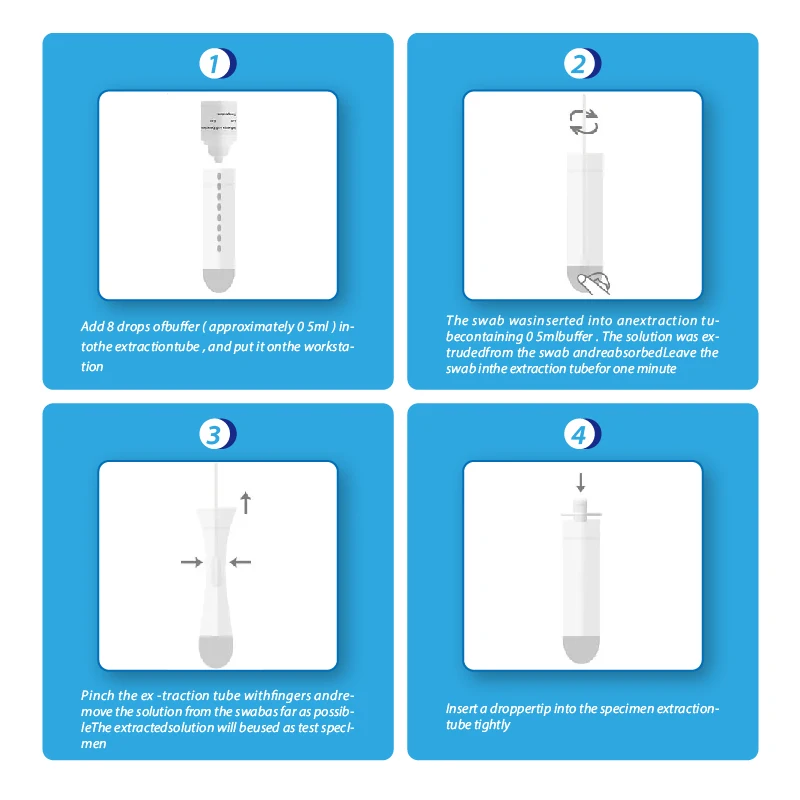
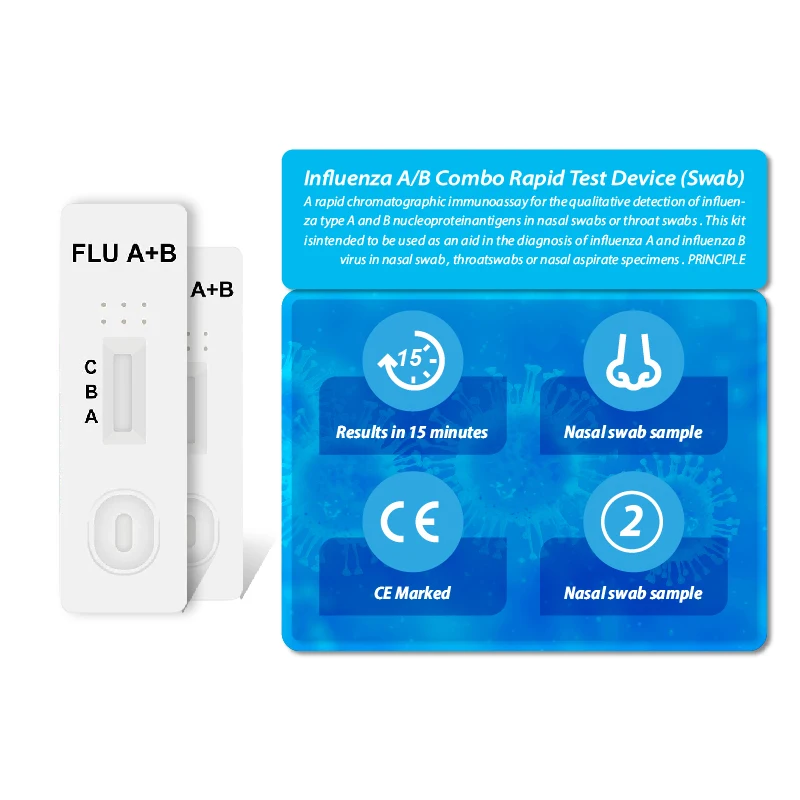
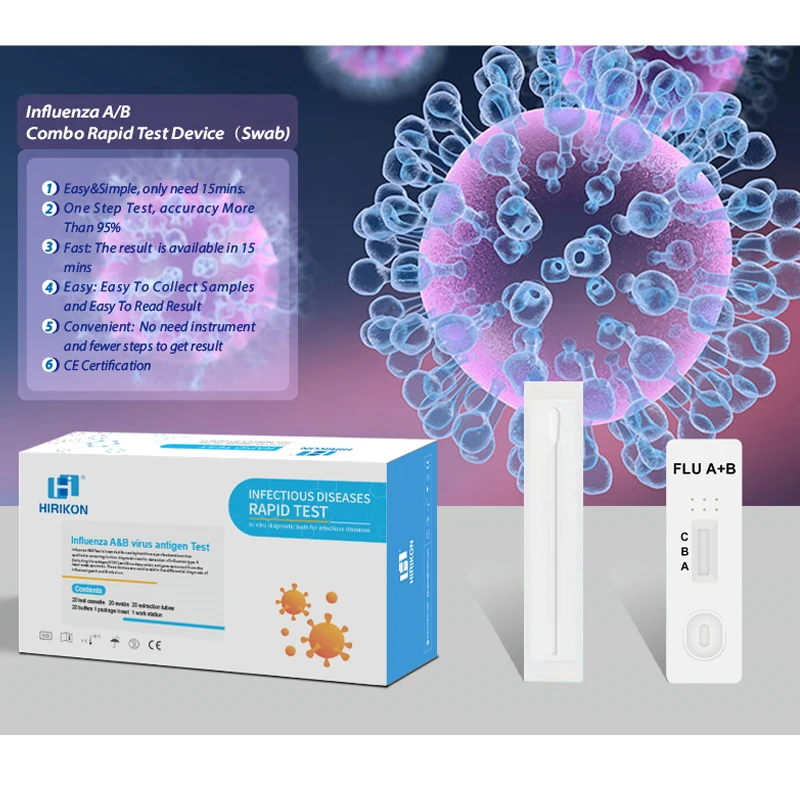
Hirikon Clinically Proven And Approved Test For Flu Virus Influenza A&B Virus Antigen Test
- Category: >>>
- Supplier: Qingdao Hihealth Medical Technology Co. Ltd.
Share on (1601003899518):
Product Overview
Description
Details Images
Products Description
The Hirikon Influenza A+B Rapid Test is a lateral flow immunoassay intended for the qualitative detection of influenza A and influenza B viral nucleoprotein antigens in nasopharyngeal swab and oropharyngeal swab from individuals with signs and symptoms of respiratory infection. It is intended to aid in the rapid differential diagnosis of influenza A and B viral infections. The test is not intended to detect influenza C antigens.
Positive results are indicative of active infection but do not rule out bacterial infection or co-infection with other viruses; clinical correlation with patient history and other diagnostic information is necessary to determine patient infection status.
Negative test results are presumptive and it is recommended these results be confirmed by viral culture or an influenza A and B molecular assay. Negative results do not preclude influenza virus infections and should not be used as the sole basis for treatment or other patient management decisions.
CAUTIONS
Please read all the data in this bundle embed prior to playing out the test.
1.For an expert in vitro indicative utilize as it were. Try not to use it after the termination date.
2.The test ought to stay in the fixed pocket until prepared to utilize.
3.All examples ought to be viewed as possibly perilous and dealt with in a similar way as an irresistible specialist.
4.The utilized test ought to be disposed of as per nearby guidelines.
5.Avoid utilizing ridiculous examples.
6.Wear gloves while giving the examples, try not to contact the reagent layer, and test well.
Company Profile
IVD-POCT Industry Leader
Hirikon is committed to providing point-of-care testing (POCT) products in the biomedical in vitro diagnostic (IVD) industry. We have 12 years of experience in medical testing and prevention and control. In 2008, Hirikon began to focus on the diagnosis and treatment. Products and services include rapid detection of colloidal gold, nucleic acid diagnosis, and Chinese medicine prevention and treatment.
Hirikon has passed various professional certifications such as CE/TUV/EUV. Overseas warehouses in Hong Kong and Europe can guarantee fast delivery.
Certifications
* ISO 13485:2016 Certified
* WHO Pre-qualified
* Follows all Good Manufacturing Practices (GMP)
* Quality Control (QC) Testing Capabilities
* Individual Component Pouching and Sealing
* Storage and Inventory of Finished Components or Final Packaging
* Advantageous logistics and shipping services
* Local, Experienced, Stable Labor Pool
Exhibition
FAQ
Q1: Are you a manufacturer? A: Yes, we are professional manufacturer with 12-years experience, 1000 workers, 40000 square meters and large production capacity. Q2: What certificates do you have? A: We have following certificates: CE, ISO13485,FSC,GMP,etc. Q3: What is your lead time? A: Normally 7-21 days, depends on your order quantity Q4: Whatis the MOQ? A: Small quantity is available like 2000pcs. Q5: Can you provide free samples? A: Yes, free samples are available. Q6: How can you guarantee the quality? A: We have mature technical team and quality department, will do inspection on every manufacturing step.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices