Wholesale price TOXO Toxoplasma Antibody Test IgM/IgG Tri line test Medical diagnostic test kit
- Category: >>>
- Supplier: Changchun ZYF Science And Technology Co. Ltd.
Share on (1601021046704):
Product Overview
Description
Product Description



Specification
Product Name
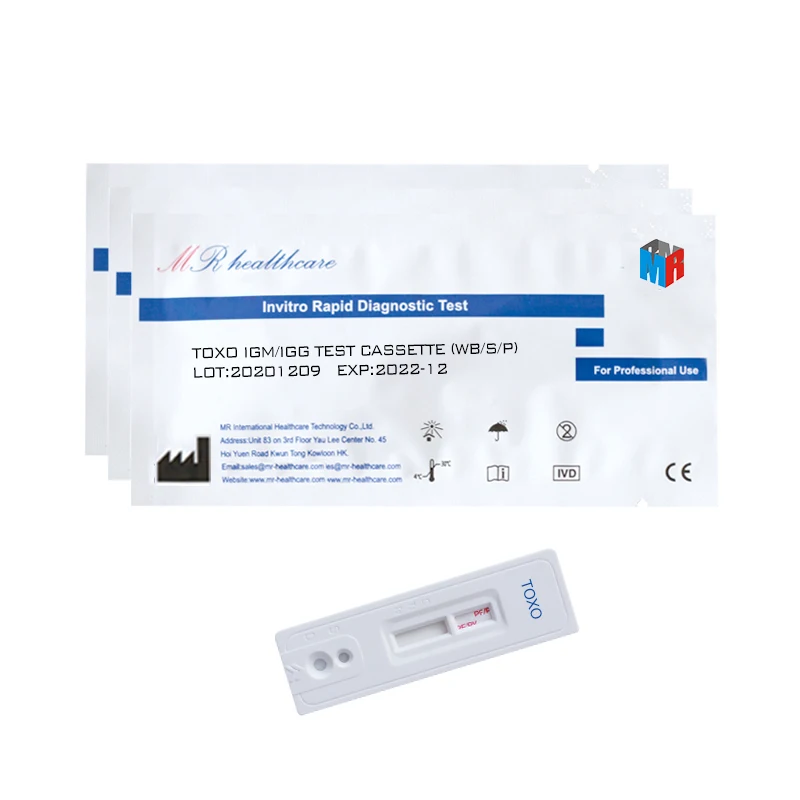
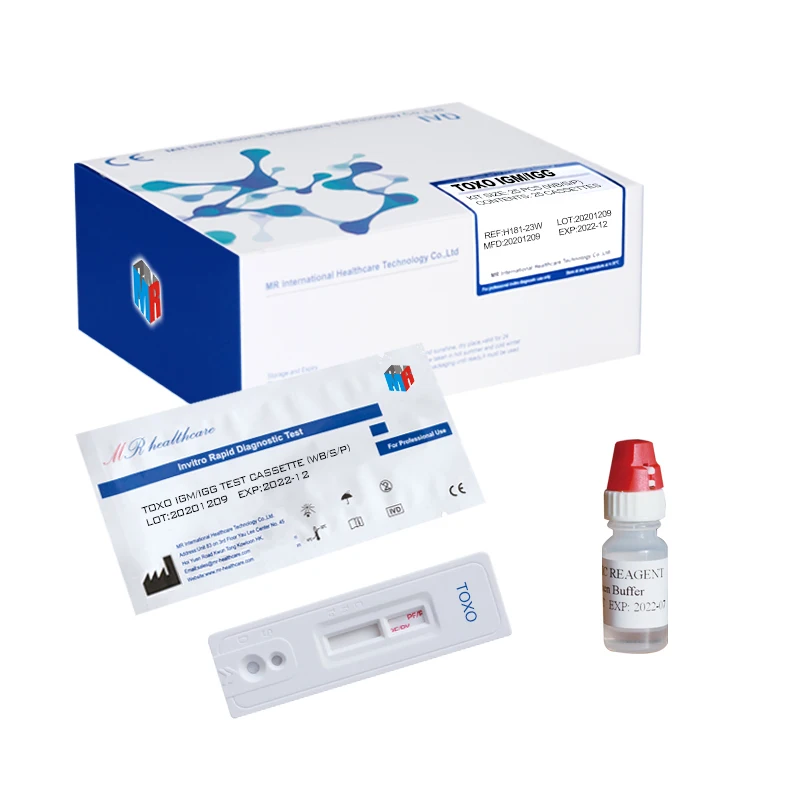
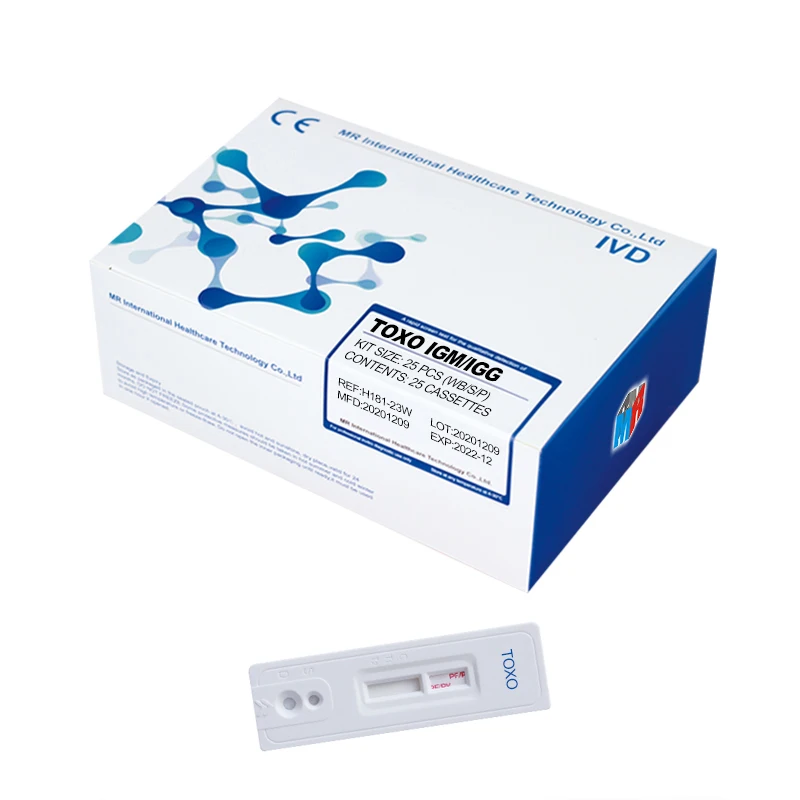
TOXO Toxoplasma Antibody Test IgM/IgG Tri-line test (Immunochromatography)
Intended Use
Used for the qualitative detection of Toxoplasma IgM and IgG antibody in human serum or plasma to determine if someone is infected by toxoplasma.
Principle
IgG antibody to TOX:
The test utilizes antibodies including a recombinant TOX antigen and goat anti-mouse IgG antibody on the nitrocellulose membrane with colloidal gold marked anti-human IgG as a mark tracer. The reagent is used to detect the TOX-IgG according to the principle of capture method and gold immunochromatography assay. The sample mixing up anti-human IgG–marker move along the membrane to the T line, and form the T line with recombinant TOX antigen when the sample contains TOX-IgG, which is a positive result. Conversely, it is a negative result.
IgM antibody to TOX:
When the sample to be detected contains the TOX -IgM antibody, it firstly forms complexes with gold monoclonal antibody, and move forwards with the effect of chromagography, then reacts with the recombinant TOX antigen. If there appears a red line (Test Line, T) which can be seen with naked eye, that is a positive result; the unreacted marker moves continuously forwards, and combines with anti-mouse antibody to form a red contrasting line(C), if the test line does not appear, that is a negative result. Main Components
Main Components
TOX-IgM: Sample pad, colloidal gold labeled anti-human IgM, nitrocellulose membrane coated with recombinant TOX antigen and goat anti-mouse IgG antibody, absorbent paper and PVC board.
TOX-IgG: Sample pad, colloidal gold labeled anti-human IgG, nitrocellulose membrane coated with recombinant TOX antigen and goat anti-mouse IgG antibody, absorbent paper and PVC board.
Storage and Expiry
Store as packaged in the sealed pouch at 4-30°C, keep out of hot and direct sunlight, keep in dry place, valid for 24 months. DO NOT FREEZE. Some protective measures should be taken in hot summer and cold winter to avoid high temperature or freeze-thaw. The product is humidity-sensitive and should be used immediately after being opened.
Care should be taken to protect the components of the kit from contamination. Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
TOXO Toxoplasma Antibody Test IgM/IgG Tri-line test (Immunochromatography)
Intended Use
Used for the qualitative detection of Toxoplasma IgM and IgG antibody in human serum or plasma to determine if someone is infected by toxoplasma.
Principle
IgG antibody to TOX:
The test utilizes antibodies including a recombinant TOX antigen and goat anti-mouse IgG antibody on the nitrocellulose membrane with colloidal gold marked anti-human IgG as a mark tracer. The reagent is used to detect the TOX-IgG according to the principle of capture method and gold immunochromatography assay. The sample mixing up anti-human IgG–marker move along the membrane to the T line, and form the T line with recombinant TOX antigen when the sample contains TOX-IgG, which is a positive result. Conversely, it is a negative result.
IgM antibody to TOX:
When the sample to be detected contains the TOX -IgM antibody, it firstly forms complexes with gold monoclonal antibody, and move forwards with the effect of chromagography, then reacts with the recombinant TOX antigen. If there appears a red line (Test Line, T) which can be seen with naked eye, that is a positive result; the unreacted marker moves continuously forwards, and combines with anti-mouse antibody to form a red contrasting line(C), if the test line does not appear, that is a negative result. Main Components
Main Components
TOX-IgM: Sample pad, colloidal gold labeled anti-human IgM, nitrocellulose membrane coated with recombinant TOX antigen and goat anti-mouse IgG antibody, absorbent paper and PVC board.
TOX-IgG: Sample pad, colloidal gold labeled anti-human IgG, nitrocellulose membrane coated with recombinant TOX antigen and goat anti-mouse IgG antibody, absorbent paper and PVC board.
Storage and Expiry
Store as packaged in the sealed pouch at 4-30°C, keep out of hot and direct sunlight, keep in dry place, valid for 24 months. DO NOT FREEZE. Some protective measures should be taken in hot summer and cold winter to avoid high temperature or freeze-thaw. The product is humidity-sensitive and should be used immediately after being opened.
Care should be taken to protect the components of the kit from contamination. Do not use if there is evidence of microbial contamination or precipitation. Biological contamination of dispensing equipment, containers or reagents can lead to false results.
Limitation
1. This reagent is designed for the qualitative screening test. Concentration of analytes cannot be determined by this qualitative test.
2. The results of the reagent are only for clinical reference, which is not the only basis for clinical diagnosis and treatment. A confirmed diagnosis and treatment should only be made by a physician after all clinical and laboratory findings have been evaluated.
3. Negative result may occur when detecting short-term infected samples, indicate that the specific antibodies of TOX does not exist or the concentration is below detection limit. If TOX infection is still suspected, the sample should be collected 2 weeks later and carry the parallel detection with the first sample.
4. Negative results may occur at the beginning of acute infection, other testing method and analysis with clinical symptoms are suggested.
5. Results of patients who used to receive immunosuppressive therapy or with immune function damage (for example HIV), may have a low serology reference value.
6. Positive results of the patients who used to receive blood transfusions or other blood products therapy, should be analyzed cautiously.
1. This reagent is designed for the qualitative screening test. Concentration of analytes cannot be determined by this qualitative test.
2. The results of the reagent are only for clinical reference, which is not the only basis for clinical diagnosis and treatment. A confirmed diagnosis and treatment should only be made by a physician after all clinical and laboratory findings have been evaluated.
3. Negative result may occur when detecting short-term infected samples, indicate that the specific antibodies of TOX does not exist or the concentration is below detection limit. If TOX infection is still suspected, the sample should be collected 2 weeks later and carry the parallel detection with the first sample.
4. Negative results may occur at the beginning of acute infection, other testing method and analysis with clinical symptoms are suggested.
5. Results of patients who used to receive immunosuppressive therapy or with immune function damage (for example HIV), may have a low serology reference value.
6. Positive results of the patients who used to receive blood transfusions or other blood products therapy, should be analyzed cautiously.
Manufacturing procedure

Packing & Delivery





Company Profile

Established in Year 2014,CHANGCHUN ZYF SCIENCE AND TECHNOLOGY Co., Ltd. is a high-tech enterprise devoted to the R&D, manufacturing, and sales of high-quality diagnostic products including laboratory equipment and clinical diagnostic reagents.
We can offer more than 30 kinds of urine analysis strips sold well and enjoyed high reputation from both domestic and overseas markets. Based on the principle of "customer first, quality first", our products continue to cover world-wide markets with the advantages of stable performance and high accuracy.
We registered MR International Healthcare Technology Co., Ltd. in Hong Kong for overseas sales. Through MR, we also possess some manufacture facilities in Zhejiang, Guangdong and Jiangsu provinces of China in order to help reduce the cost while caring about the quality.
We are committed to provide most reliable medical devices and solutions to people in every corner of the earth. Accomplishing the mission of "maximizing the reliability of medical products" is our goal. We do care about obtaining the trust from our partners and patients. We are dedicated to innovation in the fields of Rapid Test, POCT, In-Vitro Diagnostics, and Medical Consumables, such as vacuum tubes, tube holders, etc.
We can offer more than 30 kinds of urine analysis strips sold well and enjoyed high reputation from both domestic and overseas markets. Based on the principle of "customer first, quality first", our products continue to cover world-wide markets with the advantages of stable performance and high accuracy.
We registered MR International Healthcare Technology Co., Ltd. in Hong Kong for overseas sales. Through MR, we also possess some manufacture facilities in Zhejiang, Guangdong and Jiangsu provinces of China in order to help reduce the cost while caring about the quality.
We are committed to provide most reliable medical devices and solutions to people in every corner of the earth. Accomplishing the mission of "maximizing the reliability of medical products" is our goal. We do care about obtaining the trust from our partners and patients. We are dedicated to innovation in the fields of Rapid Test, POCT, In-Vitro Diagnostics, and Medical Consumables, such as vacuum tubes, tube holders, etc.
Customer Photos





Certifications


Contact us


We Recommend
New Arrivals
New products from manufacturers at wholesale prices