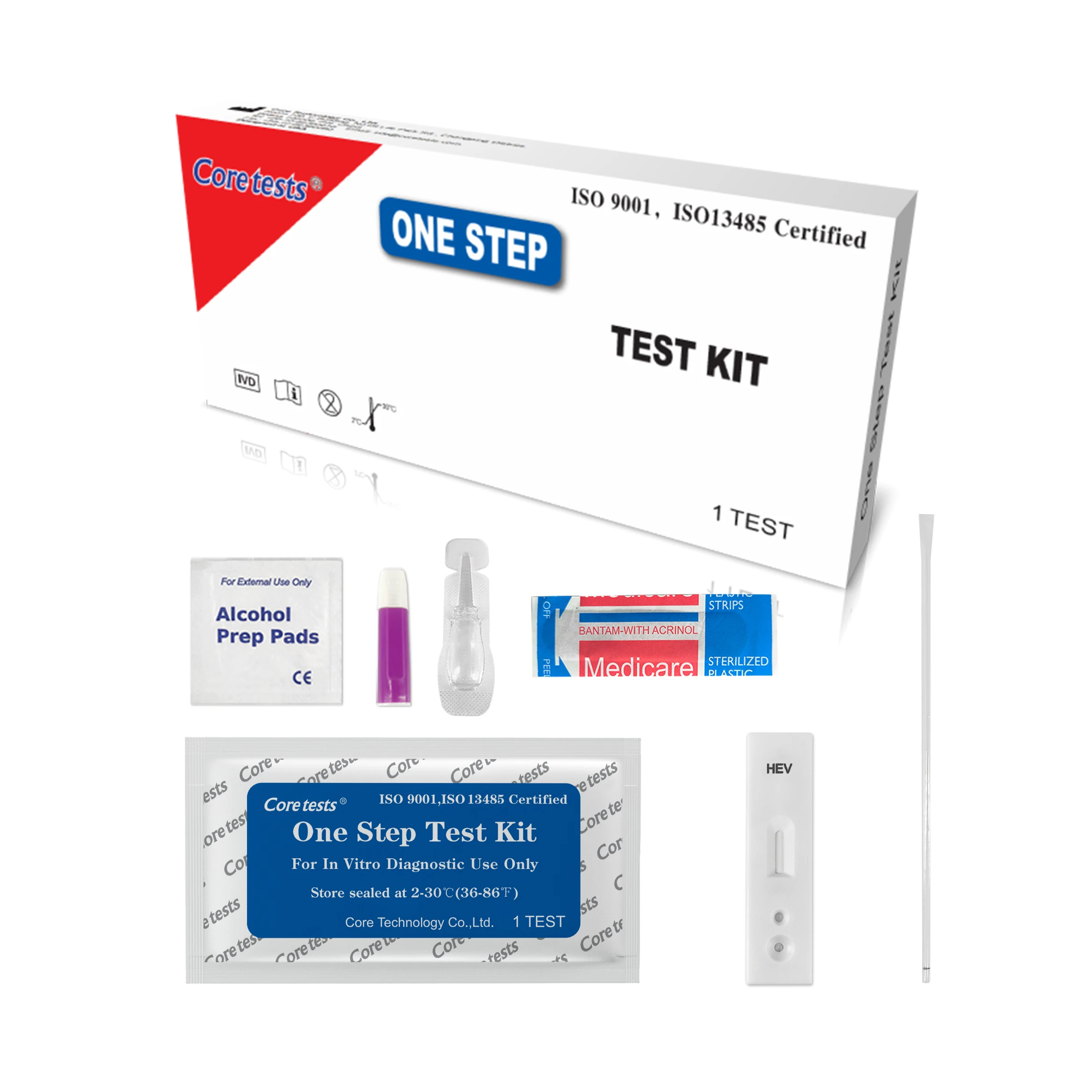
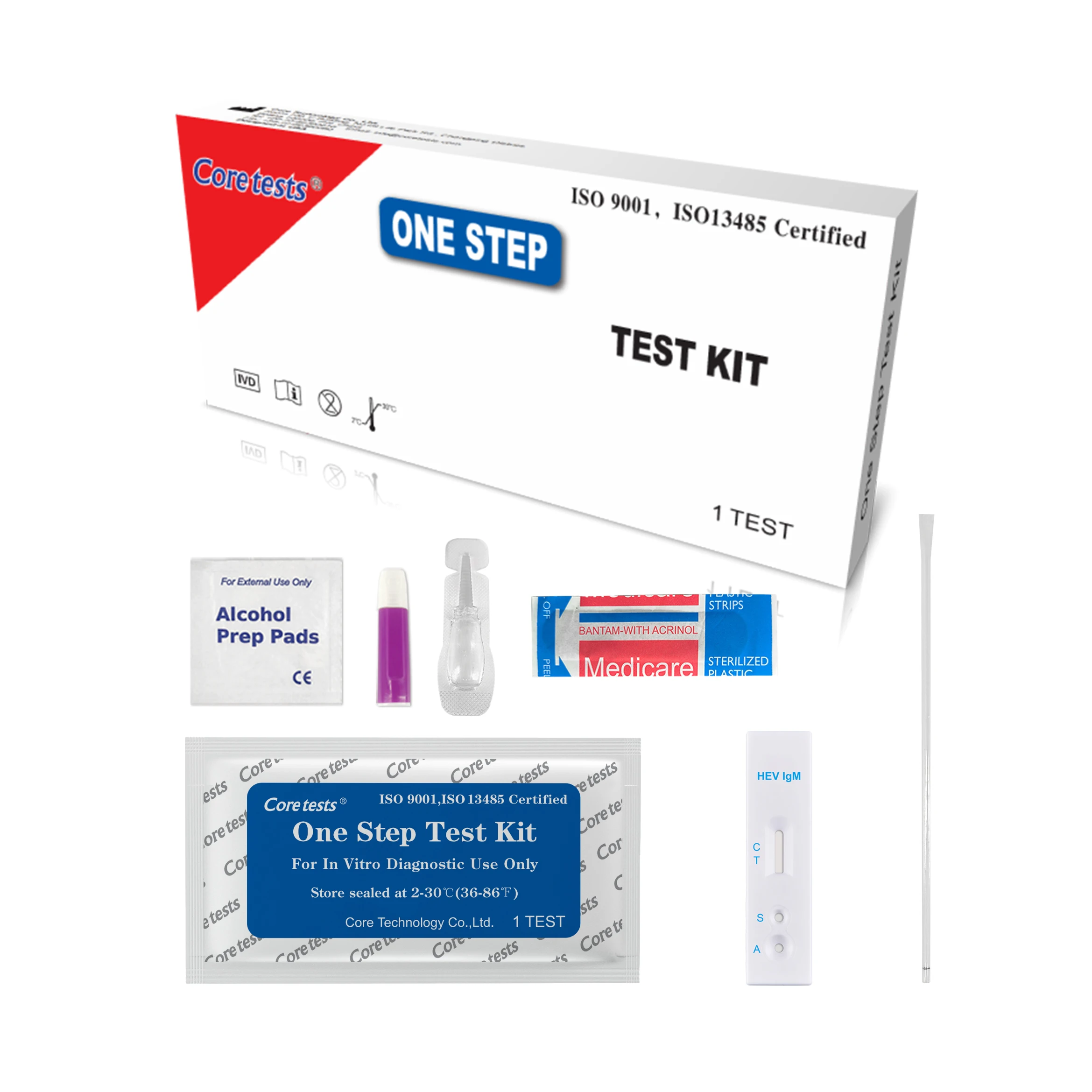
Infectious Disease Diagnostic Kit Rapid Test Device Hiv 1 2 Antigen Rapid Test Kits
- Category: >>>
- Supplier: Abiores Technology (beijing) Co. Ltd.
Share on (1601021174447):
Product Overview
Description


TEST PROCEDURE:
Bring the pouched test device to room temperature(15-30℃) prior to testing. Do not open the pouch until ready to perform the assay.
Remove the test device from the sealed pouch. Lay it on a flat, clean and dry surface.
Use the pipette to draw and slowly add 1 drop of whole blood/serum/plasma to the sample well.
Hold the buffer bottle vertically and add 1-2 drops to the sample well. If using a pipette, change a new one to avoid cross-contamination. Draw and transfer 2-3 drops of buffer to the sample well.
Interpret test results within 10-15 minutes. Do not interpret after 20 minutes.Caution: The above interpreting time is based on room temperature range of 15 - 30°C. If your room temperature is significantly lower than 15℃, then the interpreting time should be properly increased to 30 minutes.
INTERPRETATION OF RESULTS:
Negative: The presence of only one line in the control region indicates a negative result (Figure 1).
HIV-1 Positive: The control line and HIV-1 line (T1) are visible in the result window. The test is positive for HIV-1.
HIV-2 Positive: The control line and HIV-2 line (T2) are visible in the result window. The test is positive for HIV-2.
HIV-1 and HIV-2 Positive: The control line, HIV-1 (T1) and HIV-2 (T2) lines are visible in the result window. The test is positive for HIV-1and HIV-2.
Regarding the positive results for both HIV-1 and HIV-2 in one patient, it is possible for reasons as follows:
1. There is the homology in the amino acid sequence of HIV type-1 and type-2. So, it is possible that the test results show the positive results for HIV-1 and HIV-2 in one patient, simultaneously.
2. Provisionally, you can conclude virus type according to the line density. If the line density of type-1 is darker than that of type-2 in the result window, you can read as HIV-1 positive. If the line density of type-2 is darker than that of type-1 in the result window, you can read as HIV-2 positive. If you want to determine virus type or co-infection exactly, you should perform the confirmatory assay (e.g Western blot etc.).
Invalid: If the pink color line in C region is not visible, the result is considered invalid (Figure 3) regardless of the presence or absence of the test line(s).
NOTE: Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for the control line failure. Review the procedure and repeat the test with a new device. If problem persists, please contact your local distributor.
Bring the pouched test device to room temperature(15-30℃) prior to testing. Do not open the pouch until ready to perform the assay.
Remove the test device from the sealed pouch. Lay it on a flat, clean and dry surface.
Use the pipette to draw and slowly add 1 drop of whole blood/serum/plasma to the sample well.
Hold the buffer bottle vertically and add 1-2 drops to the sample well. If using a pipette, change a new one to avoid cross-contamination. Draw and transfer 2-3 drops of buffer to the sample well.
Interpret test results within 10-15 minutes. Do not interpret after 20 minutes.Caution: The above interpreting time is based on room temperature range of 15 - 30°C. If your room temperature is significantly lower than 15℃, then the interpreting time should be properly increased to 30 minutes.
INTERPRETATION OF RESULTS:
Negative: The presence of only one line in the control region indicates a negative result (Figure 1).
HIV-1 Positive: The control line and HIV-1 line (T1) are visible in the result window. The test is positive for HIV-1.
HIV-2 Positive: The control line and HIV-2 line (T2) are visible in the result window. The test is positive for HIV-2.
HIV-1 and HIV-2 Positive: The control line, HIV-1 (T1) and HIV-2 (T2) lines are visible in the result window. The test is positive for HIV-1and HIV-2.
Regarding the positive results for both HIV-1 and HIV-2 in one patient, it is possible for reasons as follows:
1. There is the homology in the amino acid sequence of HIV type-1 and type-2. So, it is possible that the test results show the positive results for HIV-1 and HIV-2 in one patient, simultaneously.
2. Provisionally, you can conclude virus type according to the line density. If the line density of type-1 is darker than that of type-2 in the result window, you can read as HIV-1 positive. If the line density of type-2 is darker than that of type-1 in the result window, you can read as HIV-2 positive. If you want to determine virus type or co-infection exactly, you should perform the confirmatory assay (e.g Western blot etc.).
Invalid: If the pink color line in C region is not visible, the result is considered invalid (Figure 3) regardless of the presence or absence of the test line(s).
NOTE: Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for the control line failure. Review the procedure and repeat the test with a new device. If problem persists, please contact your local distributor.

About us

Core Technology Co., Ltd. is a globally recognized fast growing company specializing in R&D, manufacturing, distribution and technical services of the In Vitro Diagnostic products.
With strong technical support of its R&D labs in USA and Beijing, Coretests products have been always in the frontier of innovation. Through maximizing productivity and minimizing costs for our clients, Coretests products with high performance and competitive price have been distributed in over 100 countries with hundreds of millions of users. Core Technology Co., Ltd. also specializes in customized manufacturing and contract manufacturing for many customers.
Core Technology Co., Ltd. will always stick to our faith of creating most reliable and affordable diagnostic products for people all over the world and dedicate ourselves to the health care career of all human beings.
With strong technical support of its R&D labs in USA and Beijing, Coretests products have been always in the frontier of innovation. Through maximizing productivity and minimizing costs for our clients, Coretests products with high performance and competitive price have been distributed in over 100 countries with hundreds of millions of users. Core Technology Co., Ltd. also specializes in customized manufacturing and contract manufacturing for many customers.
Core Technology Co., Ltd. will always stick to our faith of creating most reliable and affordable diagnostic products for people all over the world and dedicate ourselves to the health care career of all human beings.



Our certificate

Our advantage

Packing & shipping

We Recommend
New Arrivals
New products from manufacturers at wholesale prices