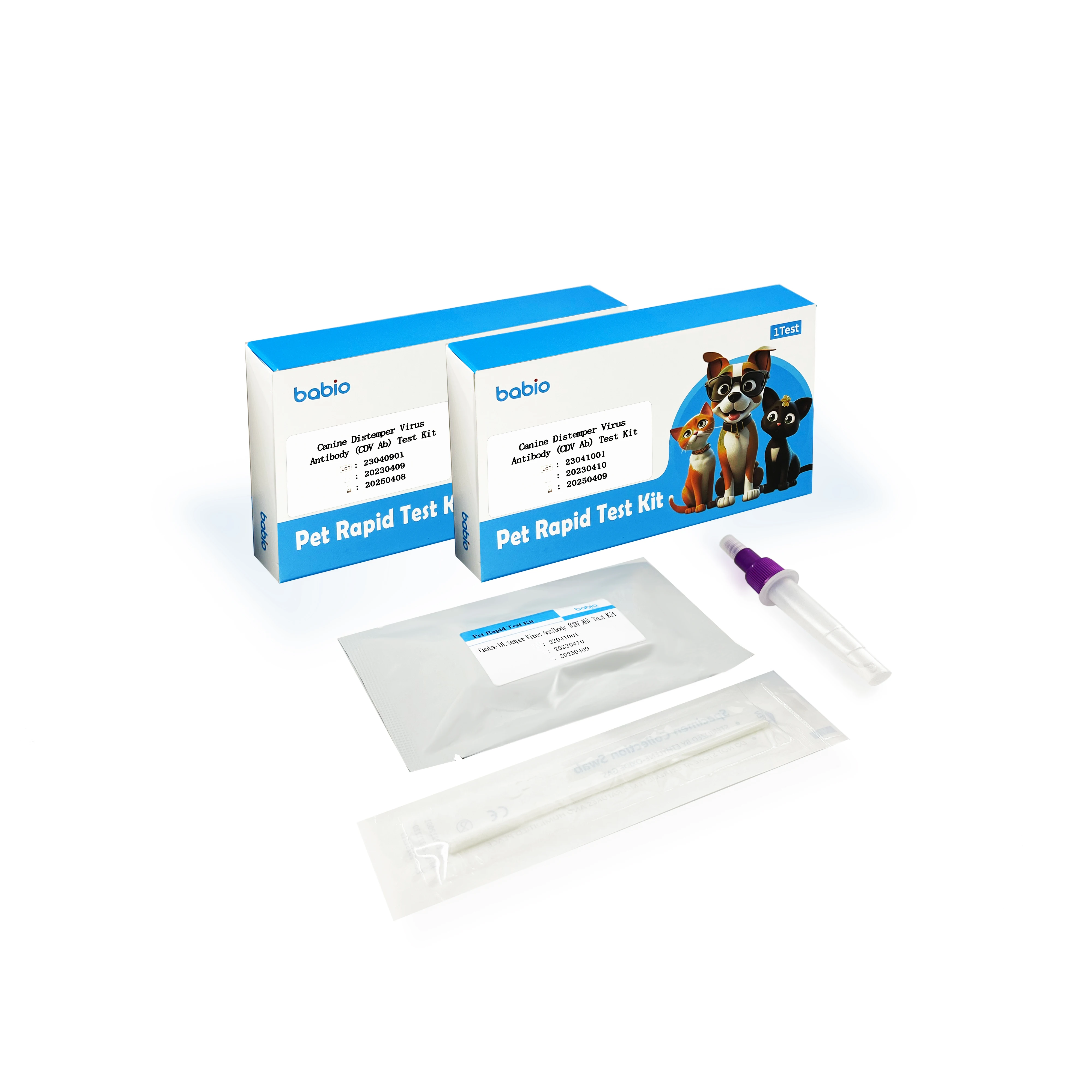
Canine Parvovirus (CPV Ag) Test Kit for Pet Dogs
- Category: >>>
- Supplier: Jinan Baibo Biotechnology Co. Ltd.
Share on (1601038937433):
Product Overview
Description
Products Description
Toxoplasma gondii antigen (TOXO Ag)Test Kit

This product can rapidly and qualitatively detect cat coronavirus antigen in cat feces, and can be used for screening and assisting diagnosis of cat coronavirus infection.
Package Specifications and Components
Components | Specification | ||||
1T/box | 20T/box | 25T/box | |||
Reagent card | 1 | 20 | 25 | ||
Diluent pipe | 1 | 20 | 25 | ||
Instruction | 1 | 1 | 1 | ||
Note: swabs are complimentary separately according to package specifications.
Test Procedure
【Sample requirement】
1. Samples: Cat eye and nose secretions.
2. Samples should be tested on the same day; Samples that cannot be tested on the same day should be stored at 2-8 ° C, and those that exceed 24 hours should be stored at -20 ° C.
1. Samples: Cat eye and nose secretions.
2. Samples should be tested on the same day; Samples that cannot be tested on the same day should be stored at 2-8 ° C, and those that exceed 24 hours should be stored at -20 ° C.

Please collect samples according to the diagram. Too many samples will result in false positive results.
Insert the above swab with a stool sample (the size of a mung bean) into the diluent tube and mix it well with the liquid. Finally squeeze the swab so that most of the solution remains in the extraction tube, then remove the swab and tighten the cap. It works best if the sample is tested immediately after collection.

【Inspection method】
1. Before use, restore the kit to room temperature (15-30℃).
2. Remove the reagent card from the aluminum foil bag and place it on a clean platform.
3. Unscrew the top tube cap on the diluent tube cap containing the sample, invert the diluent tube, squeeze the tube wall, and add 3-5 drops of specimen mixture into the sample hole (S hole) of the reagent card.
4. The results can be read in 10-15 minutes. The result is invalid after 15 minutes.

Positive: Both the quality control line (C line) and the test line (T line) appear
Negative: Only the quality control line (C line) appears
Invalid: Quality control line does not appear, take a new reagent card to re-test
Negative: Only the quality control line (C line) appears
Invalid: Quality control line does not appear, take a new reagent card to re-test
Precautions
1. This product is only used for qualitative testing and does not indicate the virus level in the sample.
2. Test results of this product are for reference only and should not be used as the sole basis for diagnosis and treatment, but should be made by a physician after evaluating all clinical and laboratory evidence.
3. A negative result may occur if the viral antigen present in the sample is below the detection limit of the assay, or if the
antigen detected at the disease stage at which the sample was collected is not present.
4. The operation should be carried out in strict accordance with the instructions. Do not use expired or damaged products.
5. The test card should be used within 1 hour after opening; If the ambient temperature is higher than 30 ° C or more humid, it should be used immediately.
6. If the T line has just begun to show color, and then the line color gradually fades or even disappears, in this case, the
sample should be diluted several times and tested until the T line color is stable.
7. This product is a disposable product. Do not reuse it.
Product display



Company Profile

Jinan baibo biotechnology co., Ltd.
Jinan Babio Biotechnology Co., Ltd. has been focusing on the R&D, production, sales and service of medical devices and in vitro diagnostic reagents since founded in 2003, aiming to provide customers with professional diagnostic solutions. Babio has passed the ISO9001 and ISO13485 certifications, is with the strict quality management leading to continuous quality improvement, has the current internationally advanced fully automatic production equipment, and possess a workshop that is designed in strict accordance with the 10,000-level purification standards.
In May 2014, Babio was successfully listed on China's NEEQ, became one of the first IVD-listed enterprises in China since the expansion of the NEEQ; In 2021, Babio successfully entered the innovation layer and became one of the few enterprises of innovation layers in the IVD field.
Babio now has a wide range of products covering cardiovascular diseases, infectious diseases, drug abuse tests, pregnancy, biochemistry, myocardial detection, laboratory consumables, and various medical consumables such as infusion, blood collection and blood transfusion, dehydrated culture media, virus sampling tubes and swabs, colloidal gold rapid detection kits, pet rapid test kits, etc. Thanks to the strong competitiveness and satisfying services, many of our products have been approved by NMPA, CE, UKCA, MDA and other international certifications, and widely sold to 100+ countries and regions, enjoying high popularity internationally.
Babio will continue being committed to building an international brand and providing users with higher-quality products with high-level technical force, strong management team, and advanced production equipment.


FAQ
Q1:Can I have sample for testing?
A:Yes, we can provide free sample for many products.
Q2: How long does shipment take?
A: We work with TNT, FedEX, UPS, EMS, DHL to supply you with the best, fastest and safest courier services. It will take about 3-10 days.
Q3:Can I add my logo on the medical products?
A:Yes,OEM and ODM are available for us. Official authorization will be required.
Q4:How can I get the after-service?
A: We will be responsible for our products during its validity.
Q5:Do you have inspection procedures for medical products?
A:We have strict QC policy under ISO system. We will conduct 100% self-inspection before packing with QC and QA team.
Q6:Can we mix the 20ft container?
A:Yes, if the items meet our min order qty.
Q7:Will you help me register the products in my country ?
A: We have professional registration team, who can help prepare certification or CTD registration dossiers if required.
A:Yes, we can provide free sample for many products.
Q2: How long does shipment take?
A: We work with TNT, FedEX, UPS, EMS, DHL to supply you with the best, fastest and safest courier services. It will take about 3-10 days.
Q3:Can I add my logo on the medical products?
A:Yes,OEM and ODM are available for us. Official authorization will be required.
Q4:How can I get the after-service?
A: We will be responsible for our products during its validity.
Q5:Do you have inspection procedures for medical products?
A:We have strict QC policy under ISO system. We will conduct 100% self-inspection before packing with QC and QA team.
Q6:Can we mix the 20ft container?
A:Yes, if the items meet our min order qty.
Q7:Will you help me register the products in my country ?
A: We have professional registration team, who can help prepare certification or CTD registration dossiers if required.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices