Pet Canine Dog Hot Cdv Cpv Parvovirus Detection Paper Test Card for Veterinary Use with the best price
- Category: >>>
- Supplier: SHANGHAI S&C BIOTECH. LTD
Share on (1601083073958):
Product Overview
Description
Introduction:
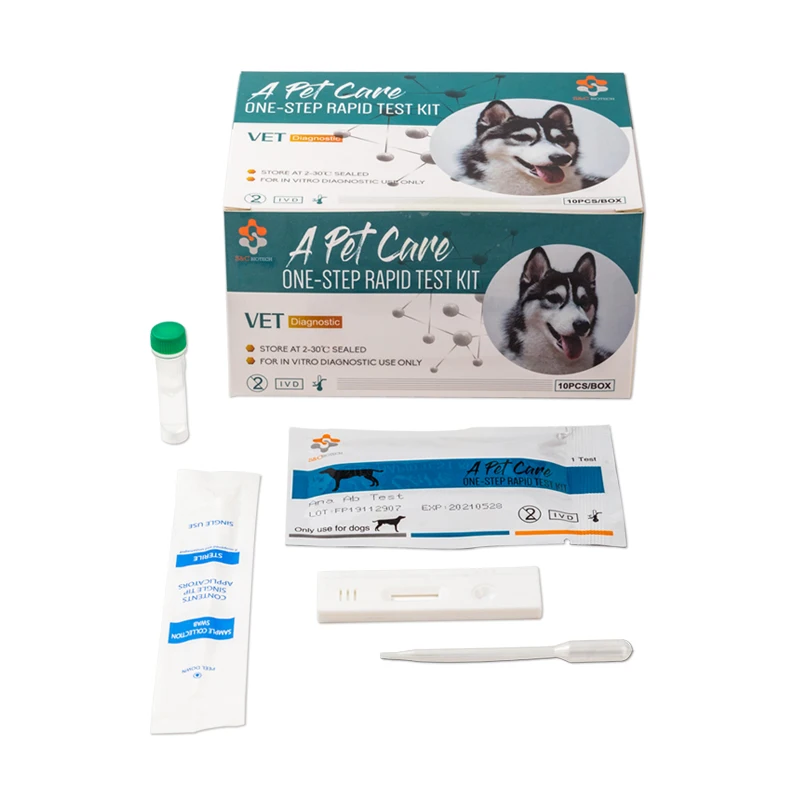
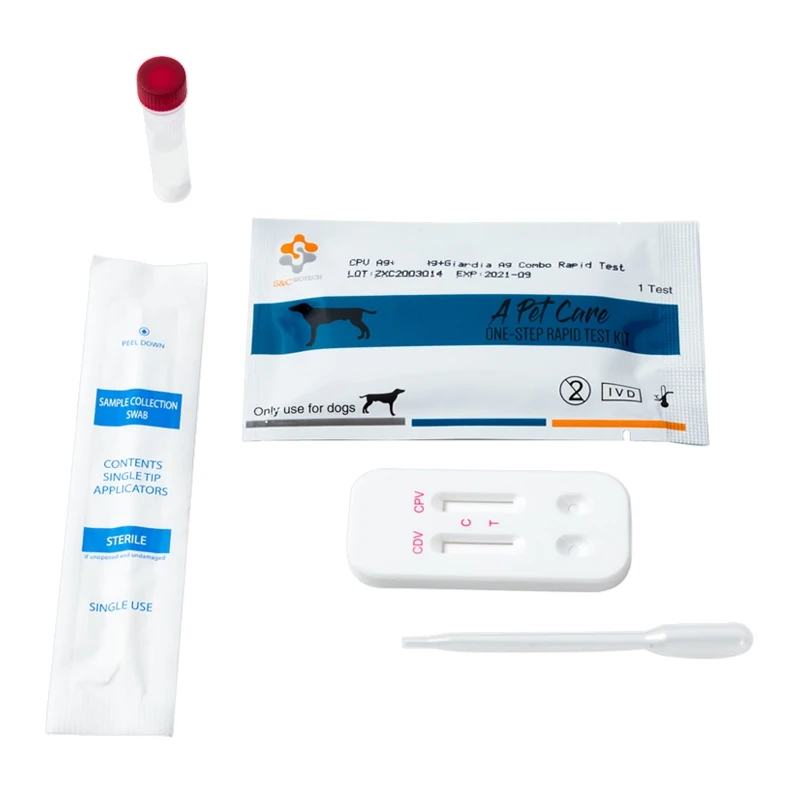
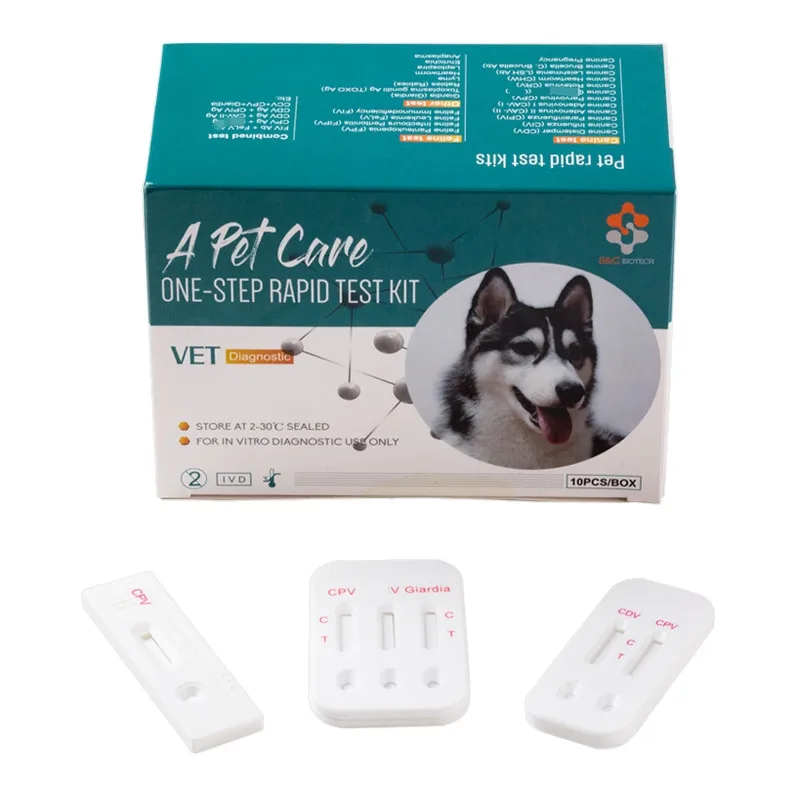
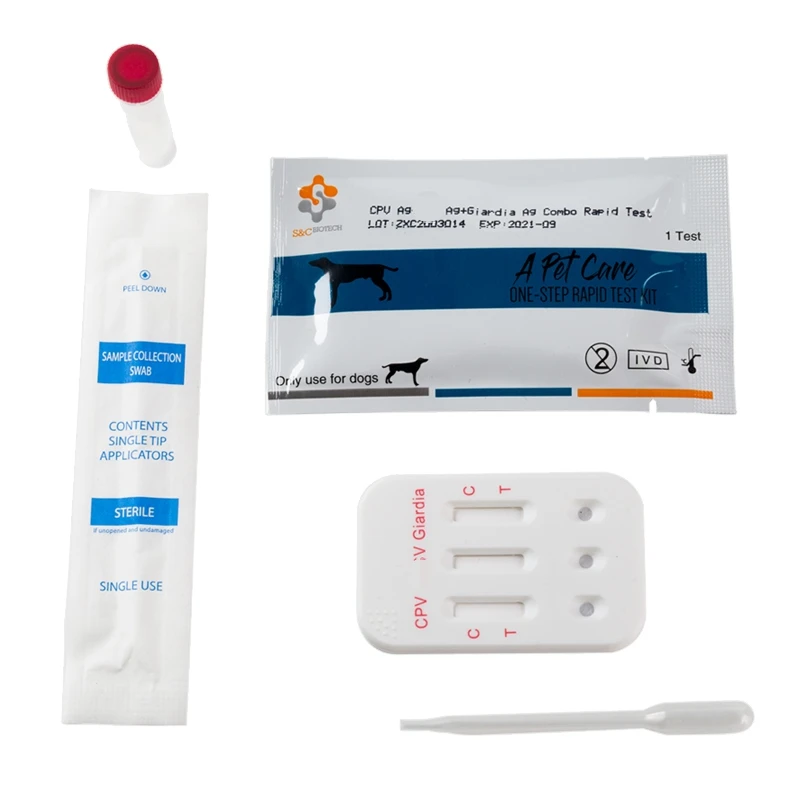
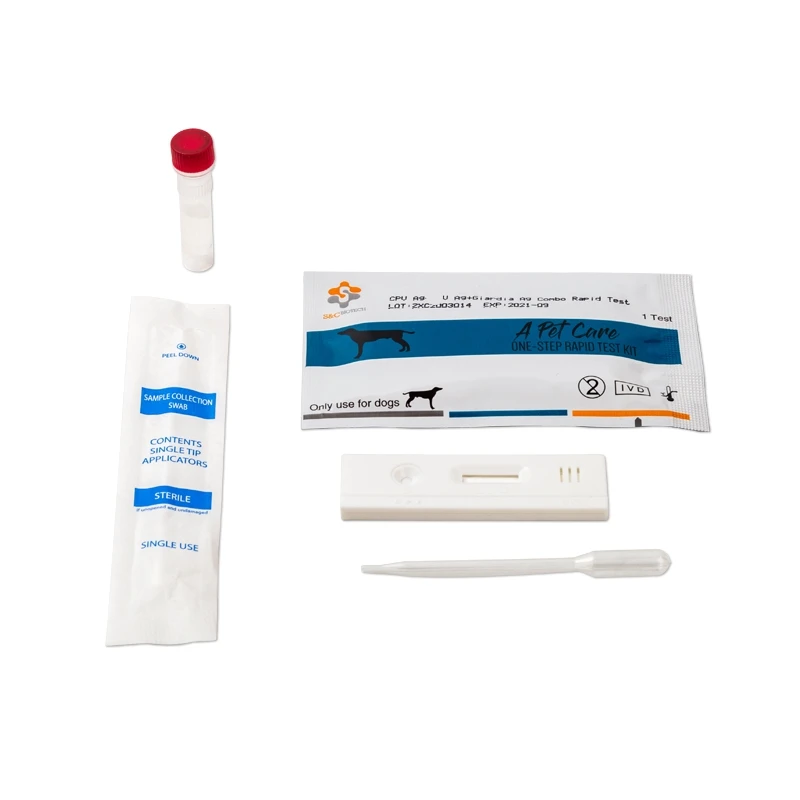
Canine Canine Distemper-Parvo Virus Antigen Rapid Test (CDV-CPV)

1. INTENDED USE
The Canine Distemper-Parvo Virus Antigen Rapid Test is a lateral flow immunochromatographic assay for the semi-quantitative detection of Canine Distemper and Parvo Virus Antigen in dog’s specimen.
Assay Time: 5-10 minutes
Specimen: CPV Ag--- feces or vomit
CDV Ag--- secretions from dog’s eyes,nasal cavities,and anus or in serum,plasma.
2. PRINCIPLE
The Canine Distemper-Parvo Virus Antigen Rapid Test is based on sandwich lateral flow immunochromatographic assay.
3. REAGENTS AND MATERIALS
- Test devices
- Disposable droppers
- CDV Ag Assay buffer (0.7mL each bottle, X10)
- CPV Ag Assay buffer (1.0ml each bottle,X10)
- Products Manual
4. STORAGE AND STABILITY
The kit can be stored at room temperature (2-30°C).
The test kit is stable through the expiration date marked on the package label.
DO NOT FREEZE.
Do not store the test kit in direct sunlight.
5. TEST PROCEDURE

CDV Ag Test Procedure
- Collect dog’s ocular, nasal or anus secretions with the cotton swab and make the swab wet sufficiently.
- Insert the swab into the provided assay buffer tube. Agitates it to get efficient sample extraction.
- Take out the test device from the foil pouch and place it horizontally.
- Suck the treated sample extraction from the assay buffer tube and place 3 drops into the sample hole “S” of the test device.
- Interpret the result in 5-10 minutes. Result after 10 minutes is considered as invalid.
CPV Ag Test Procedure
- Collect dog’s fresh feces or vomit with the cotton swab from dog’s anus or from the ground.
- Insert the swab into the provided assay buffer tube. Agitates it to get efficient sample extraction.
- Take out the test device from the foil pouch and place it horizontally.
- Suck the treated sample extraction from the assay buffer tube and place 3 drops into the sample hole “S” of the test device.
- Interpret the result in 5-10 minutes. Result after 10 minutes is considered as invalid.
6. INTERPRETATION OF RESULTS
- Positive (+): The presence of both “C” line and zone “T” line, no matter T line is clear or vague.
- Negative (-): Only clear C line appear. No T line.
- Invalid: No colored line appears in C zone. No matter if T line appears.
7. PRECAUTIONS
- All reagents must be at room temperature before running the assay.
- Do not remove test cassette from its pouch until immediately before use.
- Do not use the test beyond its expiration date.
- The components in this kit have been quality control tested as standard batch unit. Do not mix components from different lot numbers.
- All specimens are of potential infection. It must be strictly treated according to the rules and regulations by local states.
8. LIMITATION
The Canine Distemper-Parvo Virus Antigen Rapid Test is for in vitro veterinary diagnosis use only.
All result should be considered with other clinical information available with veterinarian.
It is suggested to apply a further confirmative method when positive result was observed.

Our Service:

Delivery: DHL OR FEDEX

Our Company:
Shanghai S&C BIOTECH is a professional bio-tech products manufacturer,and a Specialist in the development, manufacture, and marketing of veterinary point of care diagnostic instruments and consumables as well as animal disease diagnostic detection kits. Our aim is to supply qualified products and services to our customers for better health care .Since the establishment of our company, we have been servicing hundreds of international customers,we are working every day to better understand and address the real-world challenges faced by those who raise and care for animals in ways they find truly relevant.

We Recommend
New Arrivals
New products from manufacturers at wholesale prices