Whole Sale Price Rapid Test Kit for HBeAg, Hepatitis B Envelope test kit antigen rapid
- Category: >>>
- Supplier: Changchun ZYF Science And Technology Co. Ltd.
Share on (1601110351007):
Product Overview
Description
Product Description



Specification
Product Name
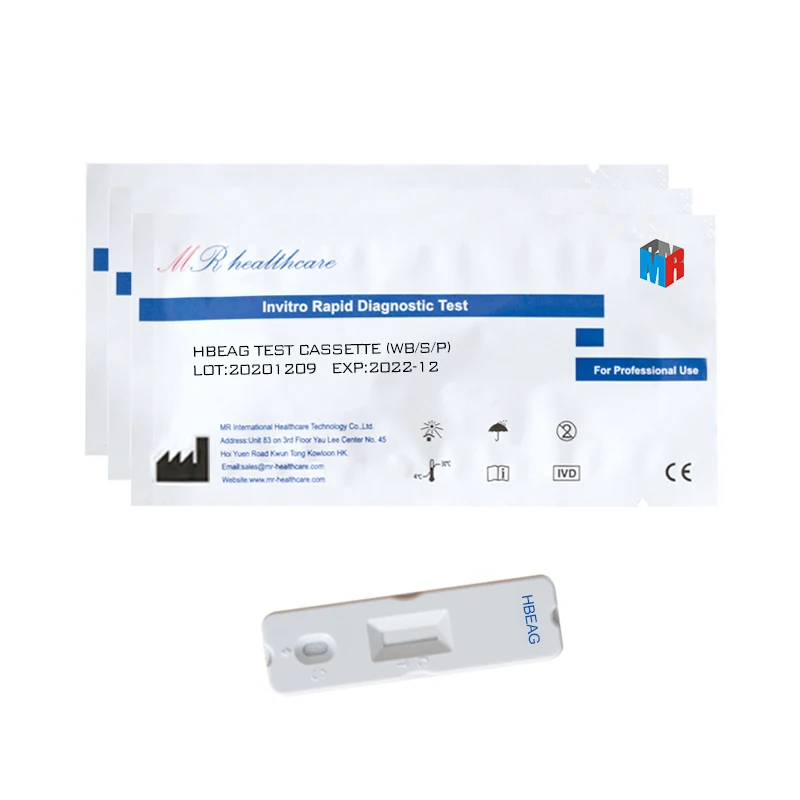
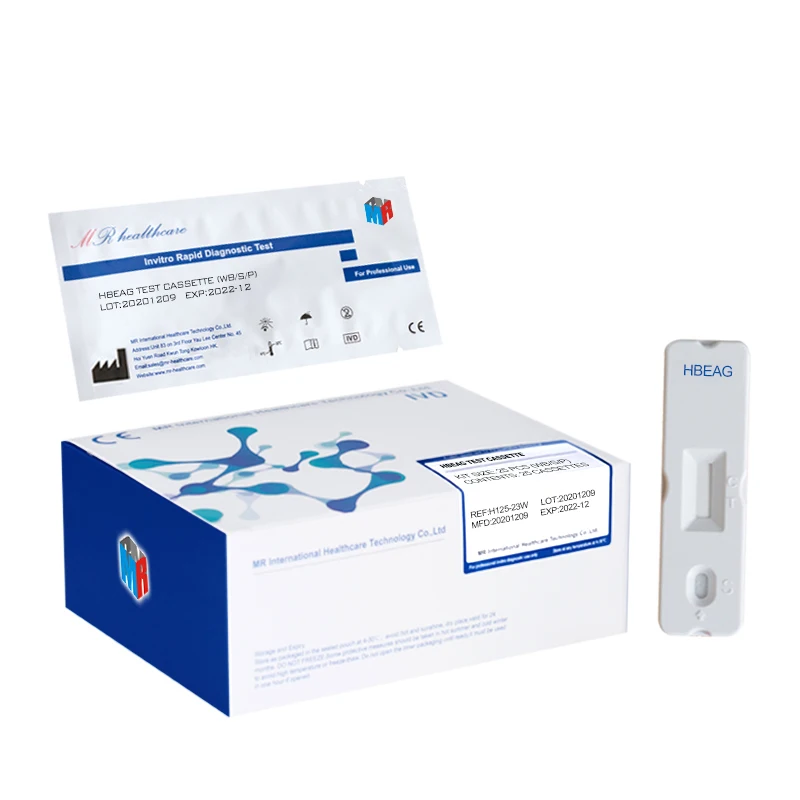
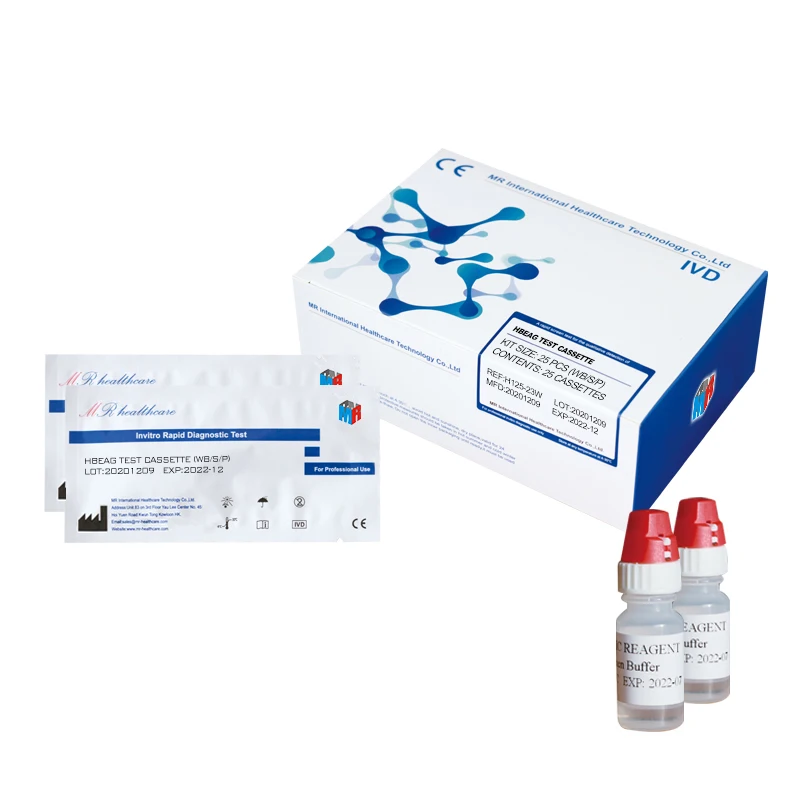
HBeAg Hepatitis B Envelope Antigen Rapid Test Kit (Immunochromatography)
Intended Use
Used for the qualitative detection of Hepatitis B Envelope Antigen in serum/plasma.
HBeAg Hepatitis B Envelope Antigen Rapid Test Kit (Immunochromatography)
Intended Use
Used for the qualitative detection of Hepatitis B Envelope Antigen in serum/plasma.
Principle
The test kit uses colloidal gold as a direct marker, uses the principle of double antibody sandwich
method for qualitative detection of HBeAg in serum/plasma.
Main Components
Sample pad, colloidal gold marked pad, nitrocellulose membrane, absorbent paper and PVC board.
Storage and Expiry
Store as packaged in the sealed pouch at 4-30°C, avoid hot and sunshine, dry place, valid for 24
months. DO NOT FREEZE. Some protective measures should be taken in hot summer and cold
winter to avoid high temperature or freeze-thaw.
The test kit uses colloidal gold as a direct marker, uses the principle of double antibody sandwich
method for qualitative detection of HBeAg in serum/plasma.
Main Components
Sample pad, colloidal gold marked pad, nitrocellulose membrane, absorbent paper and PVC board.
Storage and Expiry
Store as packaged in the sealed pouch at 4-30°C, avoid hot and sunshine, dry place, valid for 24
months. DO NOT FREEZE. Some protective measures should be taken in hot summer and cold
winter to avoid high temperature or freeze-thaw.
Sample Requirement
1. Collect venous blood a clean and dry container according to the standard method. Separate the
serum or plasma for testing. EDTA, sodium citrate, sodium oxalate, heparin can be used as the
anticoagulants
2. After collection, separate the serum and plasma as soon as possible, in order to avoid hemolysis.
(only for serum/plasma test kit)
3. Use fresh samples. Samples should be stored at -20 ℃ if cannot be tested immediately. Do not
freezeand thaw the sample repeatedly.
1. Collect venous blood a clean and dry container according to the standard method. Separate the
serum or plasma for testing. EDTA, sodium citrate, sodium oxalate, heparin can be used as the
anticoagulants
2. After collection, separate the serum and plasma as soon as possible, in order to avoid hemolysis.
(only for serum/plasma test kit)
3. Use fresh samples. Samples should be stored at -20 ℃ if cannot be tested immediately. Do not
freezeand thaw the sample repeatedly.
Limitations
1. This reagent is designed for the qualitative screening test. Concentration of HBeAg cannot be
determined by this qualitative test.
2. The results of the reagent are only for clinical reference, a confirmed diagnosis should be made
only by a physician after all clinical and laboratory findings have been evaluated.
1. This reagent is designed for the qualitative screening test. Concentration of HBeAg cannot be
determined by this qualitative test.
2. The results of the reagent are only for clinical reference, a confirmed diagnosis should be made
only by a physician after all clinical and laboratory findings have been evaluated.
Packing & Delivery





Company Profile

Established in Year 2014,CHANGCHUN ZYF SCIENCE AND TECHNOLOGY Co., Ltd. is a high-tech enterprise devoted to the R&D, manufacturing, and sales of high-quality diagnostic products including laboratory equipment and clinical diagnostic reagents.
We can offer more than 30 kinds of urine analysis strips sold well and enjoyed high reputation from both domestic and overseas markets. Based on the principle of "customer first, quality first", our products continue to cover world-wide markets with the advantages of stable performance and high accuracy.
We registered MR International Healthcare Technology Co., Ltd. in Hong Kong for overseas sales. Through MR, we also possess some manufacture facilities in Zhejiang, Guangdong and Jiangsu provinces of China in order to help reduce the cost while caring about the quality.
We are committed to provide most reliable medical devices and solutions to people in every corner of the earth. Accomplishing the mission of "maximizing the reliability of medical products" is our goal. We do care about obtaining the trust from our partners and patients. We are dedicated to innovation in the fields of Rapid Test, POCT, In-Vitro Diagnostics, and Medical Consumables, such as vacuum tubes, tube holders, etc.
We can offer more than 30 kinds of urine analysis strips sold well and enjoyed high reputation from both domestic and overseas markets. Based on the principle of "customer first, quality first", our products continue to cover world-wide markets with the advantages of stable performance and high accuracy.
We registered MR International Healthcare Technology Co., Ltd. in Hong Kong for overseas sales. Through MR, we also possess some manufacture facilities in Zhejiang, Guangdong and Jiangsu provinces of China in order to help reduce the cost while caring about the quality.
We are committed to provide most reliable medical devices and solutions to people in every corner of the earth. Accomplishing the mission of "maximizing the reliability of medical products" is our goal. We do care about obtaining the trust from our partners and patients. We are dedicated to innovation in the fields of Rapid Test, POCT, In-Vitro Diagnostics, and Medical Consumables, such as vacuum tubes, tube holders, etc.
Customer Photos





Certifications


Contact us

We Recommend
New Arrivals
New products from manufacturers at wholesale prices