Diabetic Neuropathy,Medical Equipments,Digital Biothesiometer
- Category: >>>
- Supplier: Beijing OERHUATAI Technology Co. Ltd. [Beijing China]Beijing
Share on (1624256912):
Product Overview
Description
Diabetic Market and commercial opportunity
-About 347 million people worldwide have diabetes. And 10% of diabetic patients will have diabetes complication, especially diabetes neuropathy and vascular disease (Diabetic foot disease).
-Diabetes is predicted to become the seventh leading cause of death in the world by the year 2030.
-Cardiovascular disease is responsible for between 50% and 80% of deaths in people with diabetes.
-Diabetes is a leading cause of blindness, amputation and kidney failure.
-Diabetic foot disease, due to changes in blood vessels and nerves, often leads to ulceration and subsequent limb amputation. It is one of the most costly complications of diabetes, especially in communities with inadequate footwear. It results from both vascular and neurological disease processes. Regular inspection and good care of the foot can prevent amputations. Comprehensive foot programs can reduce amputation rates by 45-85%.
So the Diabetic Market is very big.
Our near infrared therapy system could therapy diabetic foot disease and prevent amputations. It is a big commercial opportunity
Indications

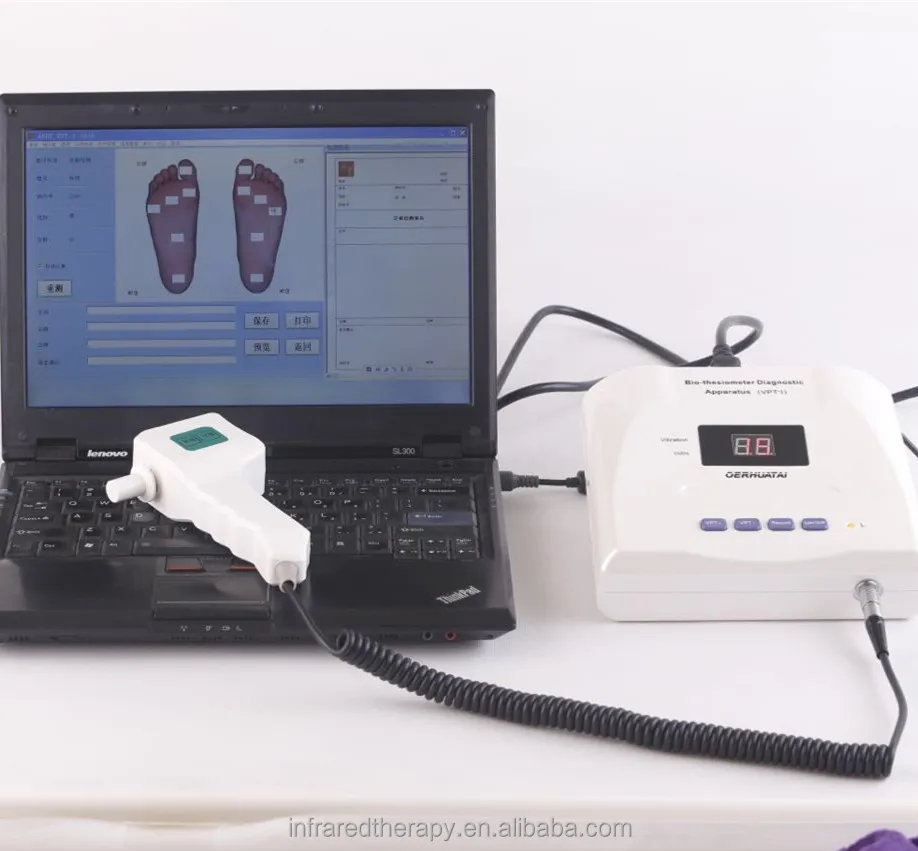
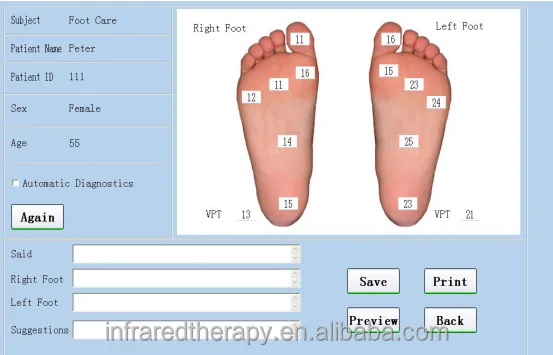
This apparatus designed to measure the threshold of appreciation of vibration in human subjects simply and accurately.
Contraindications
(1)Pregnant (including ectopic pregnancy) or patients during breast feeding period
(2)Lactational and pregnant women, hyperpietic and cardiovascular disease patients
(3)Local or systemic bacterial infection of acute period failed to controller
(4)This apparatus is not intended to be used on the cognitive disabilities
(5)The patients with psychosis and neuropathy
Work conditions
Intended use and structure chart
This apparatus is used to test the body surface neuropathy vibration perception thresholds.It could provide local vibration, and with the patient’s reaction to the induction, to auxiliarily diagnose the neural induction system diseases.

1. Digital menu board 2. Output indicated light 3. Vibration outlet;
4. Operate panel 5. Power switch 6. Output plughole;
7. Power line plughole 8. Testing handle 9. Power line.
Main Technical Parameters
(1)Operating Voltage: Alternating Current (A.C.): 220V Power Frequency: 50Hz
(2)Input Power: 50W
(3)Fuse Protector Specification: 3F1A, 250V
(4)Vibration circuit voltage range is between 0V-30V, which is continuous adjustable and stepping 0.6V, error range of ±10% .
(5)Vibration frequency of the vibration probe: 50HZ, error range of 1%
(6)The peak amplitude range of vibration probe is between 0μm -25μm, maximumpeak amplitude range of vibration probe is 25μm, error range of ± 10mm.
(7)Net weight of host apparatus: 5kg±0.5kg
(8)Size of host apparatus: 340mmx275mmx100mm, error range of ±10mm
(9)Net weight of testing handle: 0.4kg±0.05kg
(10)Size of testing handle: 170mmx90mmx40mm, error range of ±5mm
(11)Material of testing handle: ABS plastics



About Company
Beijing Huatai Healthcare Technology Co., Ltd. located in Haidian District, Beijing high-tech business park,is a high-tech enterprise and mainly engaged in research and producing medical instruments.
All the products are manufactured according to related international standards and have received universal approbation both at home and abroad. The products are already exported to Europe, America, Middle East, Africa, etc., and received widespread reputation in these countries and regions.
Contact us please
Tel:86-10-82833046 15001000273
Whatsapp: 15001000273
Website: www.huataihealthcare.com
Email: sales2(at)nearinfrared.net; maychenliu(at)126 dot com
We Recommend
New Arrivals
New products from manufacturers at wholesale prices