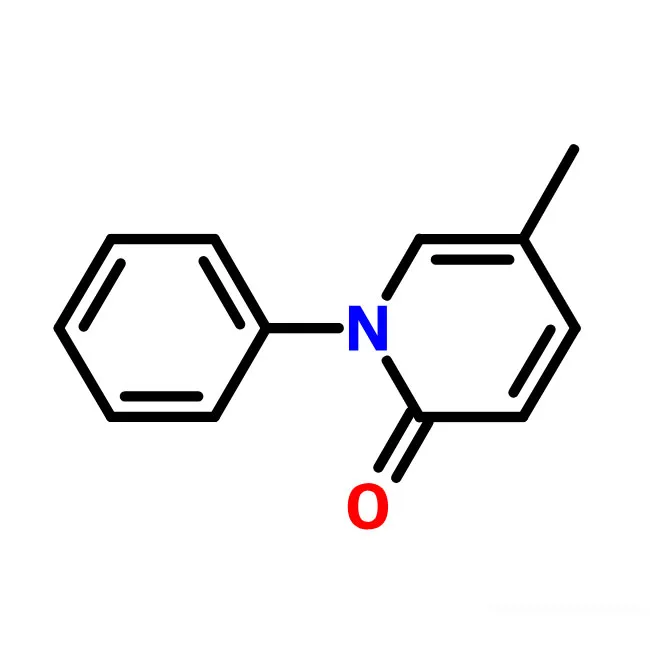
pirfenidone
- Category: Electrolyte Balance and Dialysis Agents >>>
- Supplier: Hangzhou Royall Import & Export Co. Ltd. [Zhejiang China]Hangzhou
Share on (1728568192):
Product Overview
Description
pirfenidone
The new anti-fibrotic material, with broad-spectrum anti-lung, liver, kidney, heart, and peritoneal fibrosis
| Levodropropizine |
|---|
Specification:
Items | Acceptance criteria | Results | ||
Character | Appearance: White or almost white powder. | Almost white powder | ||
Solubility: Slightly soluble in water, freely soluble in dilute acetic acid and in methanol, slightly soluble in alcohol. | Complies | |||
Identification | A.Specific optical rotation: –30.0º~-33.5º (dried substance) | –33.3º | ||
B.Infrared absorption spectrophotometry: The IR spectrum is consistent with that obtained with Levodropropizine CRS. | Complies | |||
Tests | pH | 9.2~10.2 | 9.4 | |
Impurity B and related substances | Impurity B: ≤ 0.5% | ≤ 0.1% | ||
Any other impurity: ≤ 0.1 % | ≤ 0.1% | |||
Total impurities: ≤ 0.6 % | ≤ 0.1% | |||
Impurity C | ≤ 5 ppm | ≤ 5ppm | ||
Enantiomeric purity | Impurity A: ≤ 2 % | 0.2 % | ||
Loss on drying | ≤ 1.0 %. | ≤ 0.1% | ||
Sulphated ash | ≤ 0.2 % | ≤ 0.1% | ||
Assay | 98.5% ~ 101.0%(dried substance) | 99.7% | ||
Conclusion | The product meets the specification of EP7.0 | |||





We Recommend
Greska's Carbon 60 органическое подсолнечное масло
Метформин HCL (Заводская Поставка сырья порошок)
New Arrivals
New products from manufacturers at wholesale prices