TransDetect Annexin V FITC/PI Cell Apoptosis Detection Kit
- Category: >>>
- Supplier: TransGen Biotech Co. Ltd.
Share on (60035072784):
Product Overview
Description
TransDetectTM Annexin V-FITC/PI
Cell Apoptosis Detection Kit
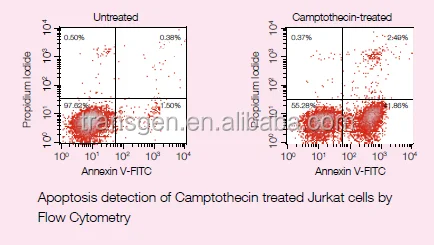
This kit can differentiate cells at different apoptosis stages when performing both Annexin V-FITC and PI staining. In a dual-parameter flow cytometry scatter diagram, if X axis indicates Annexin V signal and Y axis indicates PI signal, then the bottom left panel(Annexin V-/PI-) indicates live cells; the bottom right panel (Annexin V+ /PI-) indicates early-stage apoptotic cells; the top right panel (Annexin V+ /PI+) indicates late-stage apoptotic cells and necrosis cells; the top left panel (Annexin V-/PI+) is considered as detection error in permissible range.
Annexin V can be used as one of the most sensitive indicators to detect the early-stage apoptosis. In normal live cells, phosphatidylserine (PS) is located only on the inner face of plasma membrane. However, in the early-stage apoptosis, PS is translocated from the inner to the outer leaflet of the plasma membrane, thus exposing PS to cell surface. Annexin V is a Ca 2+ -dependent phospholipid- binding protein that has a high affinity for PS. Therefore, Annexin V can identify early-stage apoptosis by binding to PS exposed on the outer leaflet. Annexin V can be labeled with green fluorophore (FITC), the labeled Annexin V-FITC is used as a probe to detect the occurrence of apoptosis by flow cytometry or by fluorescence microscopy. Propidium iodide (PI) is a nucleic acid binding dye, and it is impermeant to live cells with intact membrane and early apoptotic cells. However, PI can stain the nucleus through permeating to cell membrane of mid-stage or late-stage apoptotic cells and necrosis cells, which show red fluorescence under certain laser excitation.
| Cat. No. | FA101 | |
| Specification | FA101-01 | 25 rxns |
| FA101-02 | 50 rxns | |
| Storage: | at 4oC in dark for one year | |
Kit Contents
Component | FA101-01 (25 rxns) | FA101-02 (50 rxns) |
Annexin V-FITC | 125 μl | 250 μl |
Propidium Iodide (PI) | 125 μl | 250 μl |
1× Annexin V Binding Buffer | 12.5 ml | 2×12.5 ml |
Procedures
1. Induce apoptosis by desired method. Collect 2×105~106 cells by centrifugation at 500×g, 4oC for 5
minutes
• For suspension cells, directly collect cells by centrifugation
• For adherent cells, treat cells with non-EDTA trypsin. After treatment, terminate reaction with serum-containing media, collect cells by centrifugation. After inducing apoptosis of adherent cells, collect the culture media as well into the centrifugation tube, centrifugate with collected cells together to ensure reliable result.
2. Wash cells twice with pre-chilled PBS, collect cells by centrifugation at 500×g, 4oC for 5 minutes.
3. Resuspend cells by adding 100 μl of pre-chilled 1×Annexin V Binding Buffer.
4. Add 5 μl Annexin V-FITC and 5 μl PI, mix gently.
5. Incubate at room temperature (20oC~25oC) for 15 minutes in the dark.
6. Add 400 μl of pre-chilled 1×Annexin V Binding Buffer. Gently mix and incubate the sample on ice in the
dark, detect by flow cytometry or fluorescence microscopy within 1 hour.
Sample Analysis
A. Analysis by Flow Cytometry Please choose appropriate voltage and adjust light compensation for flow cytometry analysis, we suggest setting control group as well.
(1) Negative control cells, no dye added.
(2) Apoptotic positive cells, single staining by Annexin V (no PI).
(3) Apoptotic positive cells, single staining by PI (no Annexin V).
Example
Jurkat cells (T-cell leukemia, human) are treated with 10 μM Camptothecin for four hours. After inducing apoptosis, follow the instruction as described above, and detect by flow cytometry, result is shown as below.

B. Detection by Fluorescence Microscopy
Place 10 μl of dual-staining (Annexin V-FITC/PI) cell suspension on a glass slide, cover the cells with a glass coverslip. Observe the cells under a fluorescence microscope using a dual filter, Annexin V-FITC shows green fluorescence, PI shows red fluorescence. Note: For analyzing adherent cells, grow cells directly on a coverslip and induce apoptosis. Then detect by fluorescence microscopy, it is better not to fix the cells. If necessary, cells must be incubated with Annexin V-FITC before fixation, in order to wash away unbound probe. For analyzing infirmly adherent cells, we suggest treating and making them into cell suspension before detection.
Notes
• Please gently handling during the whole procedure to avoid the presence of cell debris which can result in
false positive.
• Properly trypsinize cells, either insufficient digestion or excessive digestion can produce cell debris which
can result in false positive. After treatment with trypsin, ensure terminating the reaction with serum.
• Washing the cells with pre-chilled PBS cannot be omitted, and residual PBS should be removed as much
clear as possible.
• Cell samples cannot be permeabilized, otherwise AnnexinV-FITC/PI can directly enter into the
permeabilized cells, resulting in errors.
• Cell apoptosis is a constantly changing and dynamic procedure, Annexin V-FITC and PI are
photosensitive substances, thus detection should be performed as soon as possible once reaction is
completed. The whole procedure should be protected from light.
• Please centrifuge the reagents for a short time before use, in order to spin down the liquid from the tube
top and wall to the bottom.
• The successful detection of early-stage apoptosis depends on several factors, including cell status and type,
the method for inducing apoptosis and dosage, expression level of PS and the extent of its exposure to the
cell surface, the degree of mechanical damage of cell during experimental procedure. Therefore, we
suggest performing pre-experiment to optimize the corresponding steps.
• Please wear your lab coat and disposable gloves when handling to ensure your safety.



We Recommend
New Arrivals
New products from manufacturers at wholesale prices