Disposable totm av fistula needle manufacturers
- Category: >>>
- Supplier: Jiangxi Sanxin Medtec Co. Ltd.
Share on (60077323472):
Product Overview
Description
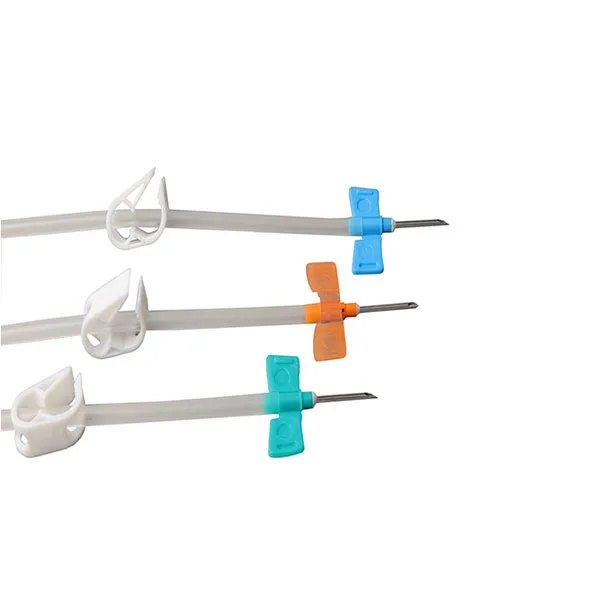
Disposable totm av fistula needle manufacturers

Product Description
Specification
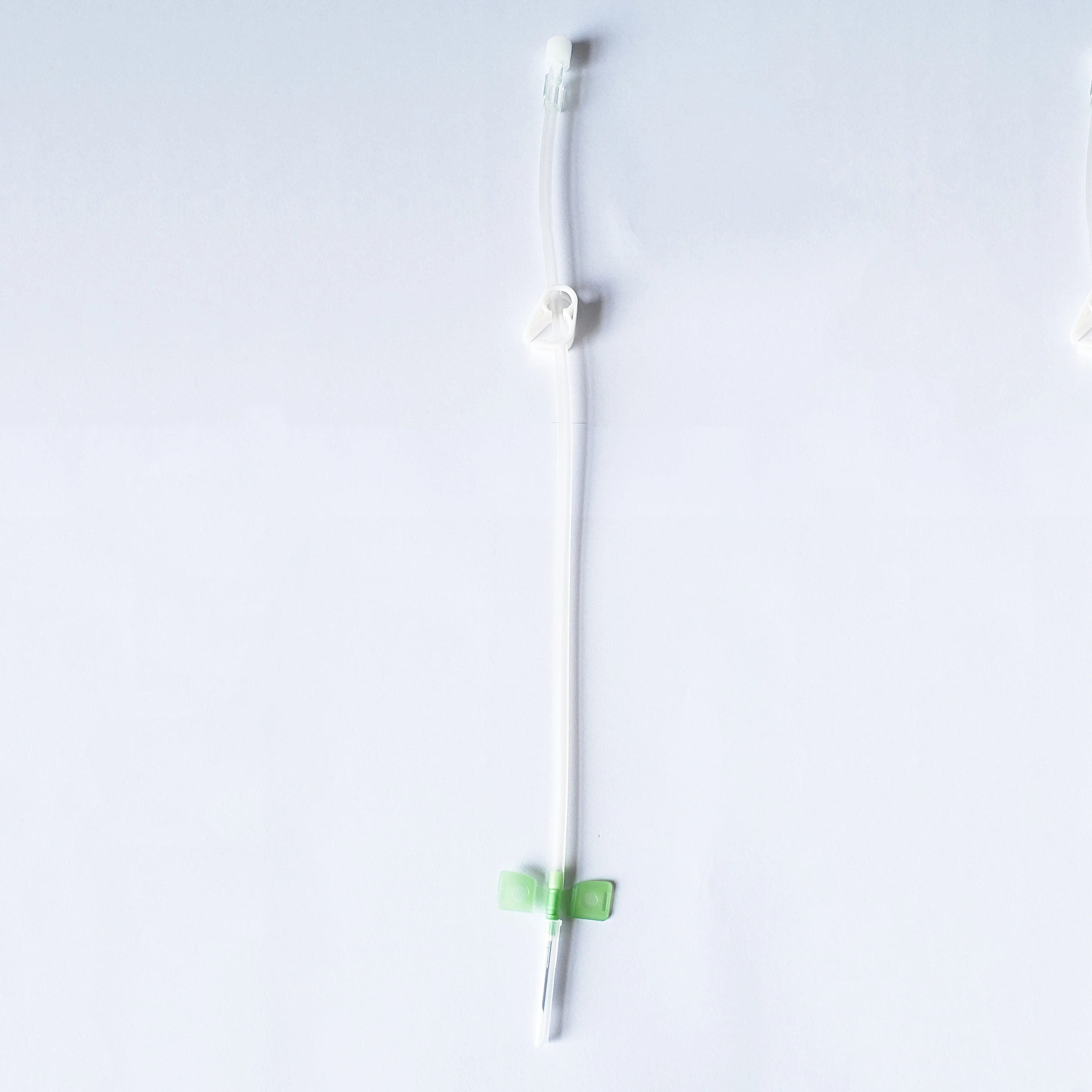
Product Picture
Safe type


Normal type

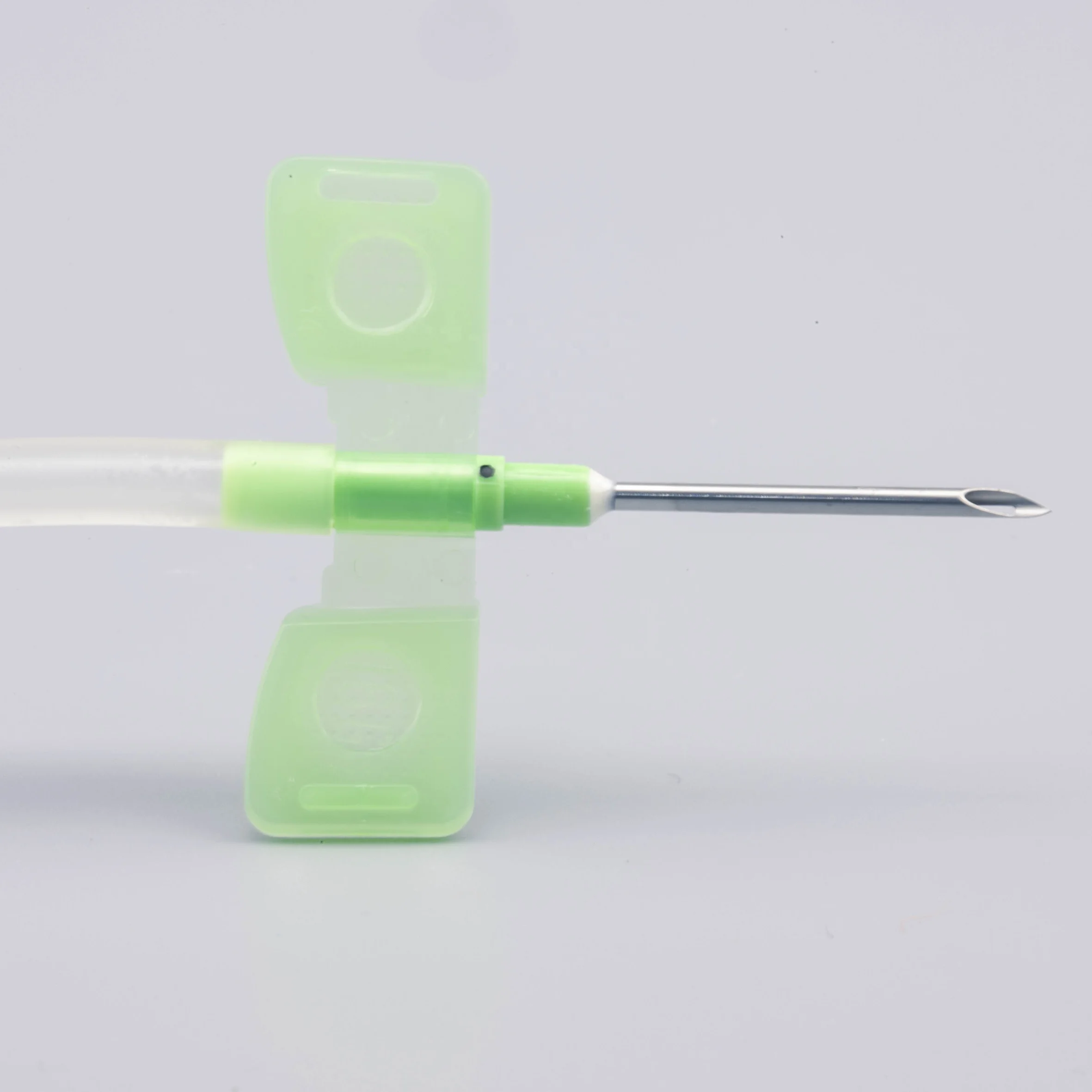
Color Code
| Type | Wing | Size | Needle tube | Color | ||
| OD | Gauge | Length | ||||
| Normal & Safety | Fixed & Rotating | 1.4 | 17G | 1"(25mm), 11/4"(32mm) | Back eye & without back eye | Yellow |
| 1.6 | 16G | Green | ||||
| 1.8 | 15G | Blue | ||||
Product Feature
1. It can be alleviate pain by the thinnest tube of A.V. Fistula needle and keep blood flow;
2. Easy to control the angle of puncture by a special concavo-convex plastic wing;
3. Soft and willowy tube can decrease the needle point's moving in blood vessel;
4. The back eye of needle point ensure plenteous blood flow; Option for fixed and rotating wing;
5. Has function of self protect, after use, could effectively protect the nurses' safety;
6. In use the protector has the function of pad, more convenient for nurses adjust the tube height effectively allay pain for patients;

Packing information
Various transportation






We Recommend
New Arrivals
New products from manufacturers at wholesale prices