Silver Silver Chloride conductive paste
- Category: >>>
- Supplier: Shenzhen Nanshan District Magaoding Chemical Material Factory
Share on (60526814047):
Product Overview
Description
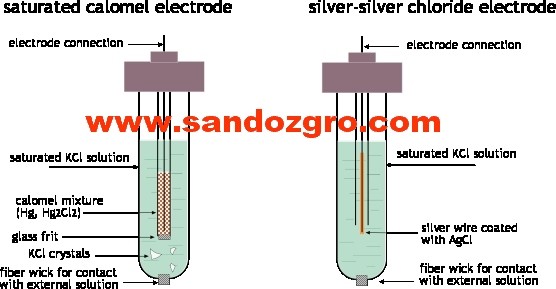
The Silver Silver Chloride Electrode paste
Basic Principles
The silver/silver chloride electrode is a reference electrode of the second kind where the metal (silver) is in contact with a slightly soluble salt (silver chloride) containing the metal, which in turn is in contact with a solution containing the common anion (chloride ion). The ions in the solid phase and those in the liquid phase are in equilibrium.
Ag solid = AgCl solid = Cl solution
The potential of the electrode is dependent on the concentration of the chloride ions in solution and temperature as governed by the Nernst equation.
ET = E0 - RT/nF ln Cl- (in moles/litre)
ET = Electrode Potential measured in Volts between a metal electrode (steel structure) and a reference electrode
E0 = Electrode Standard Potential
R = Universal gas constant
F = Faraday constant
T = Temperature
n = Number of moles
Cl = Chloride ions
APPLICATIONS
ECG–Electrocardiography
EEG–Electroencephalography
EOG–Electrooculography
ENG–Electronystagmography
EMG–Electromyography
GSR–Galvanic Skin Reflex
IMP–Impedance Pneumography








We Recommend
New Arrivals
New products from manufacturers at wholesale prices















