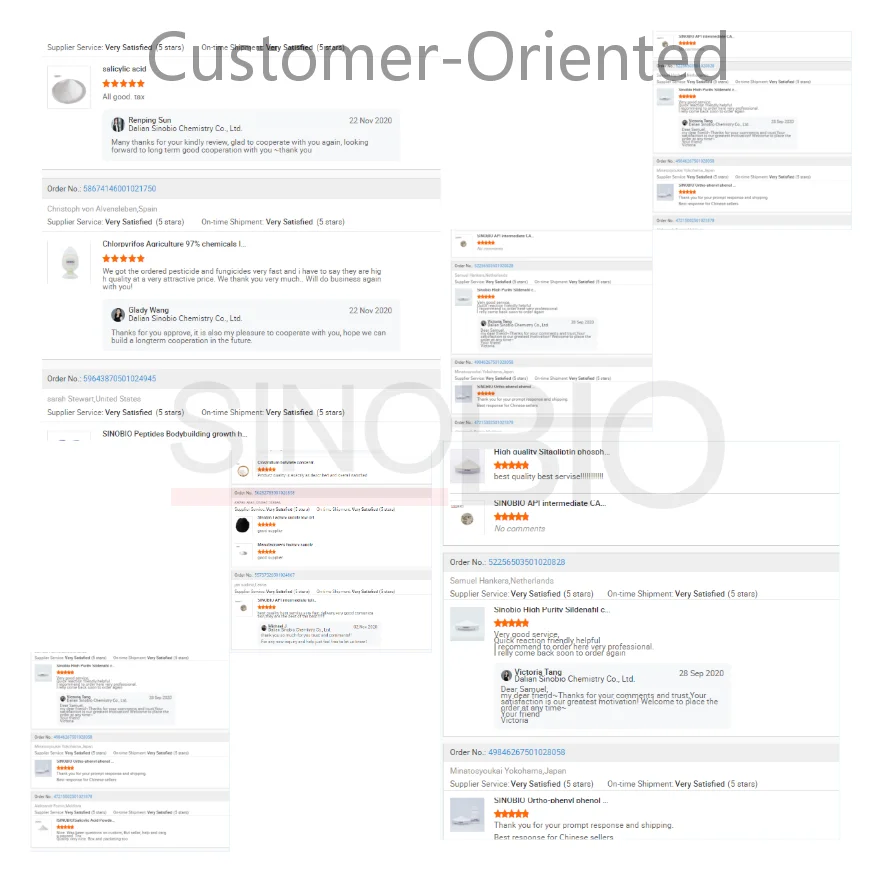
Male Sex Enhancement raw powder CAS 171596 29 5 Tadalafil
- Category: Gastrointestinal Agents >>>
- Supplier: Dalian Sinobio Chemistry Co. Ltd. [Liaoning China]Dalian Ltd.
Share on (60635969996):
Product Overview
Description
Analysis certificate of Tadalafil
|
Product |
Tadalafil |
||
|
Batch No. |
1601001 |
Date of Sampling |
March 7,2016 |
|
Quantity |
Sample |
Date of Report |
March 8,2016 |
|
Packing |
----- |
Test basis |
EP 8 |
|
Items |
Acceptance standard |
Results of analysis |
|
|
Appearance |
White or almost white crystalline powder |
White crystalline powder |
|
|
Solubility |
Practically insoluble in water, freely soluble in dimethyl sulfoxide, slightly soluble in methylene chloride |
Conform to standard |
|
|
Identification |
The infrared absorption spectrum is concordant with the reference spectrum |
Conform to standard |
|
|
The principal peak in the chromatogram obtained with the test solution is similar in retention time and size to the principal peak in the chromatogram obtained with reference solution |
Conform to standard |
||
|
Rotation |
+78.0°~+84.0° |
+80.29° |
|
|
Impurities A, B and C : |
Impurity A≤0.15% |
Non-detected |
|
|
Non specific impurity≤0.1% |
Non-detected |
||
|
Loss on drying: |
≤0.5% |
0.075% |
|
|
Residue on ignition |
≤0.1% |
0.05% |
|
|
Heavy metals |
≤0.002% |
Conform to standard |
|
|
(HPLC)Related substances
|
individual impurity≤0.1%
|
0.01% |
|
|
Total unknown impurity≤0.3%
|
0.05% |
||
|
Residual Solvents
|
methanol;≤0.3% |
Non-detected |
|
|
ethanol≤0.5% |
0.025% |
||
|
acetone≤0.5% |
Non-detected |
||






We Recommend
API-Acotiamide, High purity cas 185104-11-4 Acotiamide hydrochloride
Antiulcer drug,Sucralfate powder
PVP K30/Polyvinyl pyrrolidone/Povidone USP
Skin Cosmetic Product CAS 1239011-60-9 Acetyl Tetrapeptide-2
New Arrivals
New products from manufacturers at wholesale prices