lab urine test ovulation afp rapid test kit strip period equipment
- Category: >>>
- Supplier: Equinox Biotech Co. Ltd.
Share on (60722698076):
Product Overview
Description
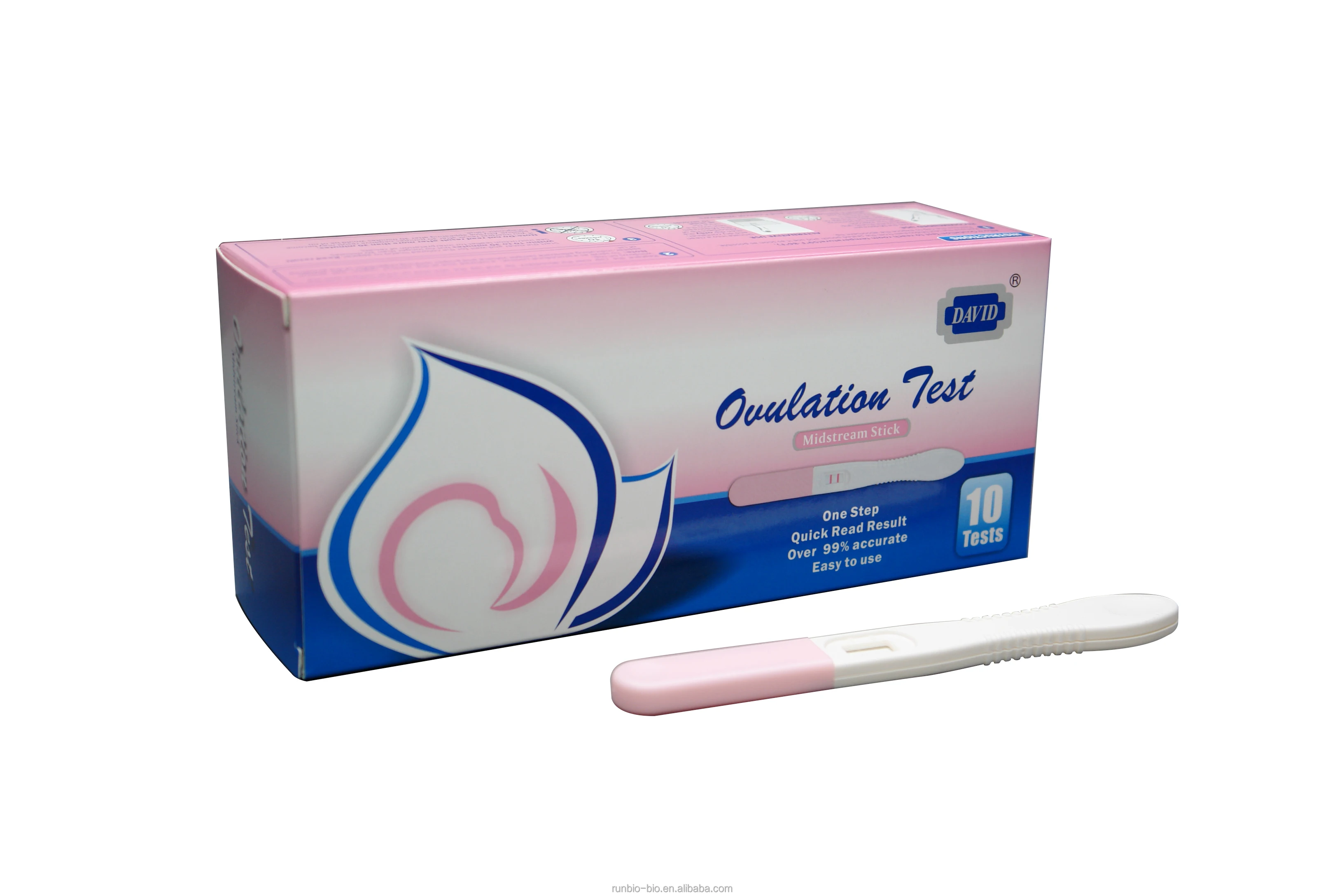
Product name | LH Ovulation test |
Sensitivity | 25 miu/ml |
Accuracy | more than 99% |
Shelf time | 24-36 month |
OEM time | 15-35 days |
Certificate | CE/ISO13485 |
Specimen | Urine |
Format | Strip /Cassette /Midstream |
Size | Strip: 2.5mm/3.0mm/3.5mm/4.0mm/5mm |
| Cassette :3mm/4.2mm |
| Midstream:4mm/6mm |
Usage | Read results within 10-15 minutes |
Sample | Sample free, freight collect |



Test Method

1. When you are ready to start the test, open the foil pouch by tearing along the notch. Take the test from the pouch and remove the cap to expose the absorbent sampler.
2. With the test pointing downwards, hold the absorbent sampler in your urine stream for 2-3 seconds (please do not stream pass the arrow end and not to splash the show window). Then you may replace the cap.1. When you are ready to start the test, open the foil pouch by tearing along the notch. Take the test from the pouch and remove the cap to expose the absorbent sampler.
3. Hold the test for a few seconds or lay the test flat. When you see the urine pass the show window and red line appear, the test is complete and has worked correctly.
4. Read result at 10 minutes but no longer than 15 minutes.
Result Interpretation
Read the result at 10 minutes but no longer than 15 minutes. A red line appears in the right sector of the result window. That is the control line, which indicates the test works properly.
* Negative: Only one red line appears in the control line or the test line is lighter than the control line. That means LH concentration is lower than detect limits and ovulation will not occur during the next 40 hours.
* Positive: If two red lines are visible and the test line is equal to or darker than the control line, it means that you will probably ovulate in the next 24-40 hours. If you want to be pregnant, the best time to have intercourse is after 24 but before 40 hours.
* Invalid: A total absence of color in both lines indicates that the result is invalid. Repeat test with a new test midstream by following the steps carefully.
Company Information
Founded in 2001, Equinox Biotech Co., Ltd. is located in the China and Israel Science and Technology Innovation Cooperation Zone (Shantou) covering an area of 20,000 square meter.
We have gotten CE and ISO 13485 certificates. Our production management is strictly in accordance with the international quality management system to ensure product quality.
Relying on the advantages of talents and technology, we have established a perfect industrialization system of R&D, production and marketing in the field of bio diagnosis, which provides a powerful driving force to the company's sustainable development.
We have world-leading production lines and experienced staffs with a daily production capacity of 2 million test kits.
Our main products are rapid diagnostic kits covering areas like fertility diagnosis, drug of abuse detection, rapid diagnosis of infectious diseases and tumor detection. At present, we have obtained 510 (k) approval, Health Canada approval and CE certification for some of the products. Because of excellent quality, such as good reproducibility and specificity, high sensitivity and easy operation, and competitive price, they are favored by customers from the United States, Europe, Africa, Middle East, Asia, South America, South Asia and other international markets.
We are committed to providing people with products and services of outstanding quality, thus making their lives easier and more convenient.



OEM Capability
EQUINOX also provides customized client services for distributors and manufacturing partners.
The production starts from raw materials and the product can be customized at any step of production. We can also provide packaging and supplies such as boxes, product shape and formats, uncut sheets, pouch printing services, and any other product modification that suits client's marketing needs. Please contact our sales and marketing team in order to obtain more information on OEM capabilities.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices