GMP Certified hip prosthesis Revision Stem Orthopedic Implant For total replacement Hip Joint
- Category: Implants & Interventional Materials >>>
- Supplier: Just Huajian Medical Devices (Tianjin) Co. Ltd. [Tianjin China]Just
Share on (60739733360):
Product Overview
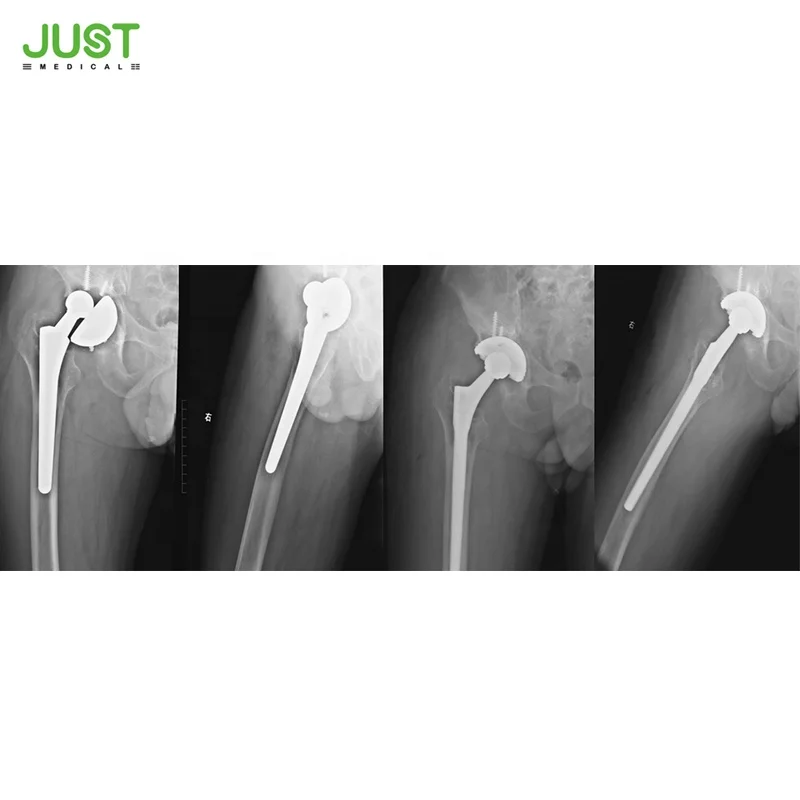
Description
GMP Certified hip prosthesis Revision Stem Orthopedic Implant For total replacement Hip Joint
Product features:
1. Making cementless fixation on the healthy bone;
2. Distal instantly anchoring on the old prosthetic bed;
3. Obtaining primary stability through press-fit effect;
4. Three flanges design strengthens the fixation of the cup, more screw choices are provided;
5. Perfect combination of rough surface is formed between bone cement and bone.
Technical parameters:
1. Material: Ti6Al4V(Stem/AOS) + Imported PE(Cup)
2. Specification: stem length 175/205/245/280, stem diameter:14-21mm.




Packaging | Inner: Three layers of sterile package, both are Tyvek + PETG heat-sealing package, covered by carton and heat shrinkable films External: Standard carton |
Lead Time | 7-21 days, To be negotiated |
Delivery | Door to door (DHL/TNT/UPS/FEDEX/EMS, etc |

HIP& KNEE Product line:


Production Show


Tests by The 3rd Party Testing Agencies
5 Million Cycles Wear Teat for TKR and THR System

10 Million Cycles Fatigue Test for Femoral Stem (Neck) and Tibial Tray

Imported Raw Material

Our service
*Prompt response to enquiry in 24 hours.
*Competitive prices and no MOQ requirement to help customers test the quality and market.
*High quality with ISO, CE certified.
*Proficient delivery and export handling.
*Surgical technique training and workshop in China and sending surgeons to demonstrate and support surgery for distributors.
*Sponsorship in advertisement and expo.
Training course for technicians of distributors CME meetings

Annual Orthopedic Meeting

We Recommend
Carbon fiber foot prosthesis implant high quality of orthopedics
Micro surgical implant titanium plate 4 holes
Titanium Skull Mesh Implant For Cranium Mesh
New Arrivals
New products from manufacturers at wholesale prices