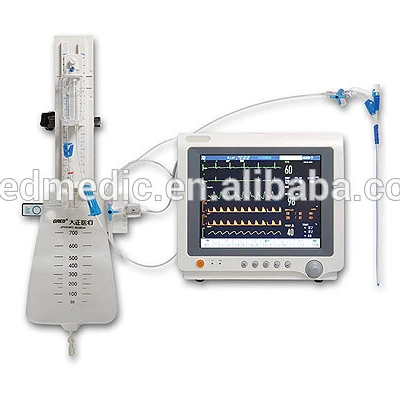
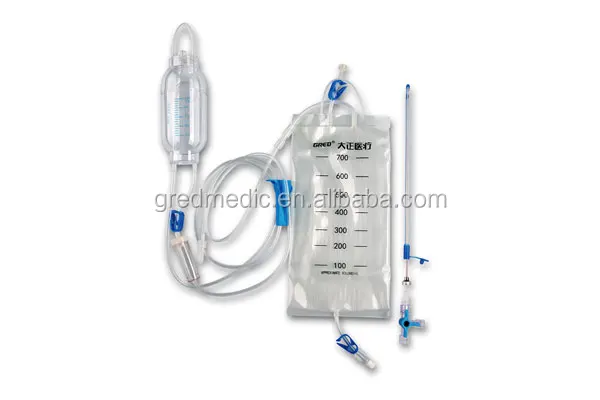
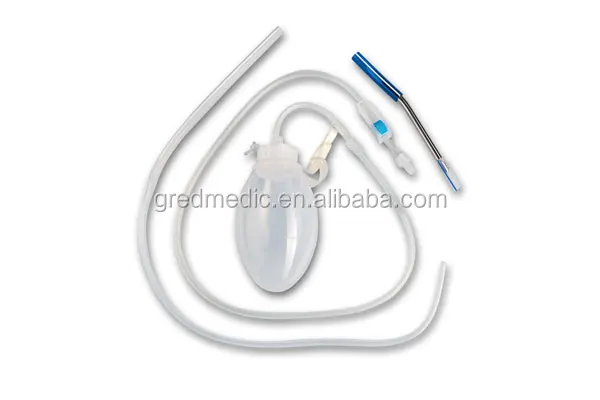
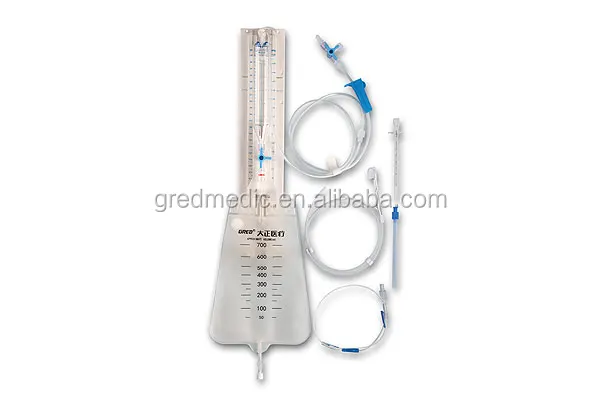
Double Channel CSF Drainage F12 Catheter System
- Category: Neurosurgery Instruments >>>
- Supplier: Shandong Gred Medic Co. Ltd. [Shandong China]Shandong
Share on (60764751439):
Product Overview
Description
Double Channel CSF Drainage F12 Catheter System

Double Channel CSF Drainage F12 Catheter System
Complete domestic production with Chinese FDAS’s approve number for Ⅲ
Leading brand for Neurological External Drainage System
Recommend products for Hemorrhagic cere stroke during Chinese twelfth five-year plan
Type; A+C+D
1. Application: Neurological External Drainage(trauma, surgery, hematoma, cerebrospinal fluid, hygroma )
2. Features for Dial
1. Accurate laser position
2. Centimeter water column and millimeter mercury (HO2 and Hg)
3. Inspect the intracranial pressure
4.Press bottle adjusted up and down frequently and well firmed
5.Double Air filtration membrane
6.Anti-reflux drainage bag, with hydrophobic air filter membrane
7.Imported Superior triple-valve
8.anti-backflow, anti-siphon, Anti-infection
Double channel catheter features:
1. Imported silicon material
2. Washing as well as fluid replacement, it can work for infection and samples.
Nurosurgical External Drainage
Infant Care Kit
ICP Monitor and Consumable
With 37million registered capital, Shandong GRED Medic CO., LTD (hereinafter referred as GRED) was founded in 1995 in Weihai, a beautiful coastal city. GRED is a Provincial Level high-tech enterprise who is specialized in R&D, production and sales of medical devices. The major products include: neuro drainage devices, ICP monitors, puncture devices, surgical devices in Department of Neurosurgery, consumables and supportive accessories in Department of Obstetrics and Gynecology and Surgery.
GRED owns a total of 36 patents: 3 invention patents and 33 utility model patents and only in the field of neuro-surgery, the patents we have already received are 27. Class III product of “disposable brain external drainage device” was named third prize in Municipal Science and Technology Progress Award. The joint project “Soft channel---- External Drainage Minimal Invasive Treatment of Cerebral Hemorrhage” was promoted as a key project nationally by Chinese Medical Doctor Association in 2007 and it was followed by 36 training sessions provided by GRED. In 2012, the neuro drainage device was appointed as a recommended product by Hemorrhagic Stroke and Minimal Invasive Treatment Technology Research Group under the “12th Five-Year” state technology supporting plan.
The manufacturing factory covers an area of more than 40,000 square meters, and owns 8,000 square meters for the 100,000-class GMP clean workshops.
High-standard central laboratory is equipped with all kinds of inspection equipments, which can inspect raw materials and finished products to guarantee quality of all products. GRED strictly follows quality management system requirements and has received the certifications of ISO13485:2003 quality system in medical device industry and ISO9001-2000 quality system.

1. - Q:Are you a factory or trading company?
- A: We are a factory, we have rich experience for manufacturing more than 10 years.
2. - Q:Where is your factory located?
- A: Our factory is located in Weihai city,Shandong Province,China.
3. - Q:Can you provide OEM for us?
- A: Yes, OEM is fine.
4. - Q:Can you send samples?
- A: Yes, samples are available.
5. - Q:How does your factory do as quality control?
- A: Quality is the lift of our factory. We have inspector in different post, and there are many inspectors before packing to get best quality products.
6. -Q: Which exhibition will you participate in this year?
- A: We will participate in the China Canton Fair and CMEF in May, the Medica Exhibition in November. We will show our exhibition number after we get it, and hope to meet you there.
Contact Information
We Recommend
New Arrivals
New products from manufacturers at wholesale prices