exact perfect accurate hba1c analyzer for POC reagent
- Category: >>>
- Supplier: Shijiazhuang Hipro Biotechnology Co. Ltd.
Share on (60768839548):
Product Overview
Description
[Product Name]
General Name:Glycosylated Hemoglobin (HbA1c) Test Kits(Nephelometry immunoassay Method).
Trade Name:Hipro™ HbA1c Test
[Packing]
25 Tests/ Kit.
[Intended Use]
This product is applicable to the clinical determination of the content of blood for glycosylated hemoglobin, and specific reagent for the specific protein analyzer, applies only to the clinical in vitro assisted diagnosis.
Glycosylated hemoglobin is the formation of hemoglobin and a combination of sugars by non-enzymatic. The combination of process is slow and relatively irreversible, and the persistence of the life of 120 days in the red blood cells is directly proportional to the concentration of the sugar in the synthesis rate of erythrocyte surroundings. Therefore, the percentage of glycosylated hemoglobin reflects the determination of the average blood sugar levels within 1~2 months before testing.
[Principle]
The HbA1c units is the use of antigen-antibody reaction to the direct determination of HbA1c percentage of total Hb. Samples of total Hb, HbA1c and latex in the sample will solidified by the same non-specific adsorption. The latex-HbA1c-mouse anti-human HbA1c monoclonal antibody complexes formed when the HbA1c of specific monoclonal antibody joined in. This composite was agglutinates because of sheep anti-mouse lgG antibodies and will produce the phenomenon of light scattering. Content is proportional to the intensity of scattered light and samples of HbA1c. Specific protein immunoassay to measure the intensity of scattered light, and comparison with the standard curve of HbA1c percentage concentration to get HbA1c percentage of the total Hb content in the sample.
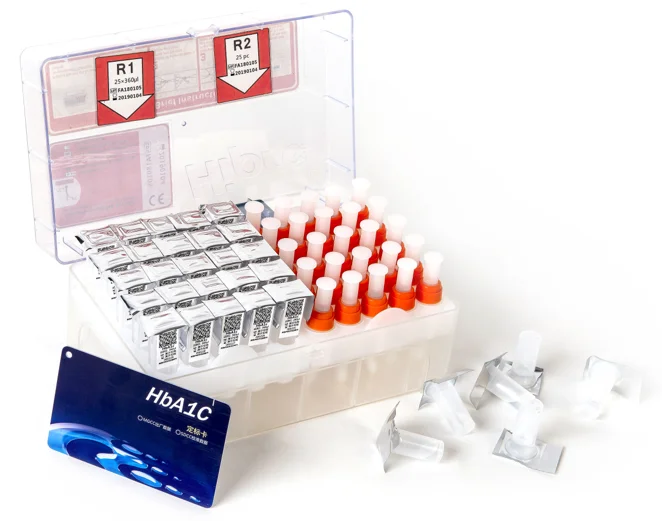
The kits contains all the reactive reagents.(figure 1)

Reagent 1 Reagent 2
[Components]
| Content | Quantity |
Reagent 1(Reaction Cup) | 15mmol/L glycine buffer | |
latex | 0.1% | |
Reagent 2 | glycine buffer | 60mmol/L |
goat anti-rat IgG, rat anit-human HbAlc monoclonal antibody |
| |
Buffer | Pure water | 0.5ml |
IC card | / | 1 |
Do not mix different batches of reagents.
[Storage and Validity Period]
Store at: 2~8℃.
Validity Period: 1 year.
Do not keep the kits at room temperature for long time.
[Applicable Device]
HP-083/4-I specific protein analyzer, HP-083/4-II specific protein analyzer, HP-AFS/3 specific protein analyzer, HP-AFS/1 specific protein analyzer.
[Specimen]
Whole blood, finger blood or venous blood, anticoagulation including EDTA, heparin, and citrate.
Anticoagulation, mixing 10μL whole blood sample with 0.5mL purity water . The sample store at 4℃ in dark place for 10 days, Avoid repeated freezing.
[Procedure]
Preparation
The operation of specific protein analyzer please refer to the instruction. Start up the analyzer 30 minutes before the test.
Attention:·
HP-083/4-I,HP-083/4-II Specific protein analyzer: insert the IC card into the slot, press the corresponding channel button, input the parameter information.
Procedure
HP-083/4-I,HP-083/4-II Specific protein analyzer:
1. Sample Preparation: The test kit is equilibrated to room temperature, Add 10μL whole blood samples( finger blood or venous blood) into dilution tube filled with 0.5mL pure water by pipette or microcapillary with fully mixing.
Important Note:
Once the capillaries are full of blood samples, test immediately, do not test before fulfill the capillaries.
2. Sample Collection:
3. Sample Mixing:
4. Reagent (R2) Injection:
Inject the reagent R2 into the cuvette.
5. Reagents Mixing:
2 Attention
Mixing Time:3-5 seconds.
6. Test:
Insert into the corresponding test channel, the results will be displayed on the window and printed automatically.
HP-AFS/1,HP-AFS/3 Specific Protein Analyzer:
1. Sample Preparation: The test kit is equilibrated to room temperature, add 10μL whole blood samples( finger blood or venous blood) into dilution tube filled with 0.5mL pure water by pipette or microcapillary with fully mixing.
Important Note:
Once the capillaries are full of blood samples, test immediately,
2. Sample collection:
Take 10μL diluted samples by capillary in front of the sample collector, insert the sample collector into the cuvette.
3. Test:
Insert the R2 reagent into R1 cup, insert the R1 cuvette into the test channel, the test will be done automatically, The results will be displayed on the window and printed.
Attention:
After step 2, insert the cuvette into test channel directly. Do not mix the sample and press the piston. (As figure)
Calibration
This product can be traced back to IFCC reference materials and testing methods. The calibration values for the different lots of the kits are stored on the calibration IC card or the two-dimensional code on the cuvette. Before test the new lot of kits, read the calibration card parameters first. Or the instrument automatically scan the two-dimensional code on the cup to obtain the corresponding calibration curve during testing.
Quality Control
3- level calibration system guarantee the results' reliability for each lot of test kits, including the instrument calibration, remote reagent calibration and the third party calibration.
The third party calibration applicable for:
1. The daily indoor quality control test.
2. New lots of reagent.
3. New operator training.
4. The results can not match the clinical symptoms
5. The first use of the reagent
If still can not be calibrated, contact the manufacture for further technical support.
[Reference Value]
3.8%-5.8%
Recommended that each laboratory establish its own reference range.
[Interpretation]
The test result between 6%-7%, the blood glucose in good condition, 7%-8% in general condition,8%-9% is in bad condition, need to strengthen blood sugar control, pay more attention to diet and exercise, and adjust the therapeutic schedule guided by doctor; >9% indicates in dangerous condition, is a risk factor for the development of chronic complications, may lead to diabetic nephropathy, atherosclerosis, cataract and other complications, and there may be ketoacidosis and other acute complications.
The result only for clinical reference, comprehensive consideration should be combined with the clinical management of patients with symptoms / signs, medical history, other laboratory tests and treatment response.
All laboratory tests depend on random errors. If the test results are in doubt, or if they do not match the clinical symptoms, re-test the sample or confirm the results with other methods.
[Limitations]
Bilirubin≤648 µmol/L, triglyceride≤9.8mmol/L, NOT effect the test results.
[Performance]
1.Linearity range: 2%~14%
2. Detection limit: ≤1.2%
The limit of detection means the lowest detectable analyte level that can distinguish the concentration. Calculate based on the minimum standard above the two standard deviation of the data ( Blank table, 1+2SD, within-run precision,n=21).
About Hipro
Hipro biotechnology Corp, founded on September 29th, 2006, is a high-tech enterprise based on international advanced medical technologies and brilliant self-innovations to provide first class products all over the world. Hipro focuses on R&D, manufacturing, marketing, and relevant services of Point-of Care products. Hipro, with her R&D center in Silicon Valley US, and production base in Hebei Province, has established branches in San Francisco, Beijing, Suzhou, Chengdu, Guangzhou,Mumbai,etc., and launched 3.69 acres industrial park project in China Medical City.








Packing: We use high-strength and environmental protection material to pack our product. The packaging can protect the goods from dropping from 20 meters high place.

Shipping:



or other air transportation.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices