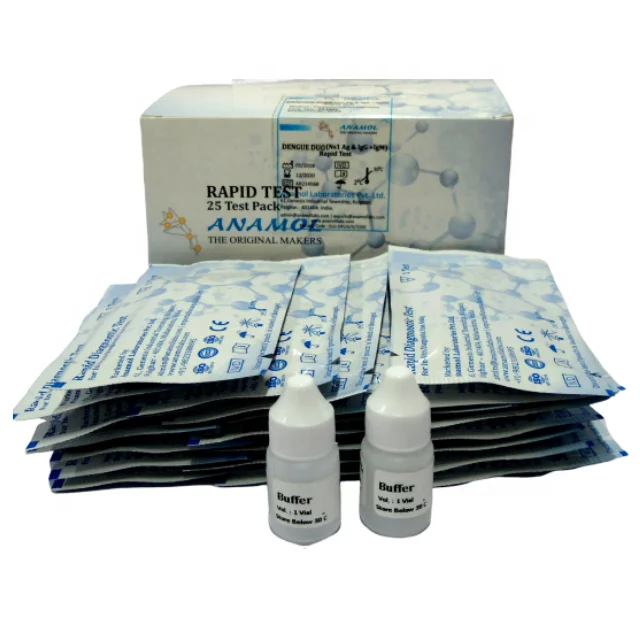
Malaria Dengue Rapid Detection Test Kit
- Category: >>>
- Supplier: ANAMOL LABORATORIES PRIVATE LIMITED
Share on (62022351915):
Product Overview
Description
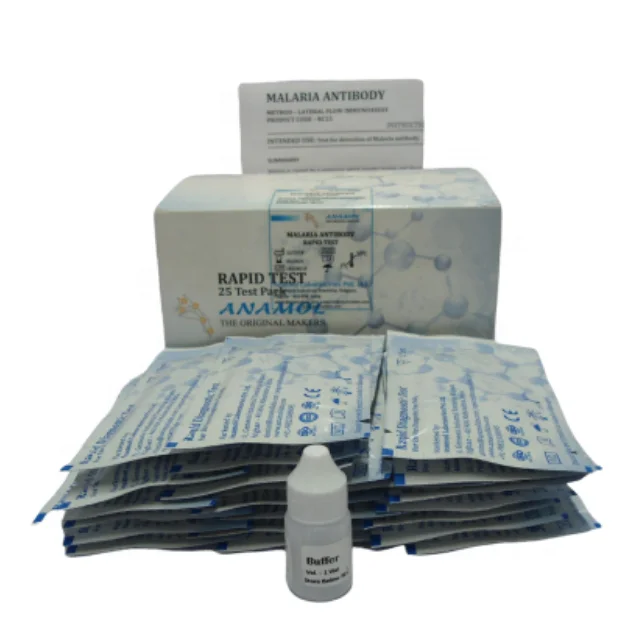
MALARIA Pf/Pv ANTIGEN CARD TEST
Intended Use:
Test for qualitative detection of Malaria P. falciparum and P. vivax in human whole blood samples.
Introduction:
Malaria is a serious parasitic disease characterized by fever, chills and anemia and is caused by a parasite that is transmitted from one human to another by the bite of infected Anopheles mosquitoes. There are four kinds of malaria that can infect humans: Plasmodium falciparum, P. vivax, P. Ovale, and P. Malariae. In humans, the parasites (called sporozoites) migrate to the liver where they mature and release another form, the merozoites.
Principle:
The Malaria Antigen Test contains a membrane strip, which is pre-coated with two monoclonal antibodies as two separate lines across a test strip. One monoclonal antibody (test line 1) is specific to the P. falciparum histidine rich protein-2 (pf HRP II) and another monoclonal antibody (test line 2) is pan specific to lactate Dehydrogenase (Pan LDH) of plasmodium species (P.Falciparum, Vivax, Malariae, Ovale). Conjugate pad is dispensed with monoclonal antibodies conjugated to the colloidal gold, which are specific to P. Falciparum histidine rich protein-2(pf HRP-II) and pan specific to the Lactate Dehydrogenase (Pan LDH) of other Plasmodium species.
DENGUE IgG/IgM ANTIBODY CARD TEST
Intended Use:
Test for detection of IgG and IgM antibody against Dengue virus in human serum and plasma samples.
Introduction:
Dengue virus, a virus belonging to the Flavavirus group of viruses, is one of the most significant mosquito-born diseases in the world in terms of morbidity and mortality. There are four known serotypes of dengue. Symptoms of dengue fever include high fever, headache, muscle pain and skin rash. The complications often associated with this infection are dengue hemorrhagic fever or dengue shock syndrome. The immune response to this virus includes the production of IgM antibodies by the 5th day of symptoms, which remain in the circulatory system for 30-60 days. IgG antibodies appear by the 14th day of infection and persist for life. A secondary infection often results in high fever and, in many cases, initiates hemorrhagic events and circulatory failure. A secondary infection also induces an IgM antibody response after 20 days of infection and IgG antibodies rise within 1-2 days after the onset of symptoms.
Method Principle:
First a specimen is dispensed with sample buffer, the Gold antigen conjugate will bind to anti-Dengue IgG and IgM antibodies in the specimen sample which in turn will bind with Anti-Human IgG and Anti-Human IgM coated on the membrane as two separate lines in the test region as the reagent move across the membrane. The anti-Human antibodies on the membrane will bind the IgG or IgM antigen complex at the relevant IgG and or IgM test lines causing pale or dark pink lines to form at the IgG or IgM region of the test membrane. The intensity of the lines will vary depending upon the amount of antibody present in the sample. The appearance of pink line in a specific test region (IgG or IgM) should be considered as positive for that particular antibody type (IgG or IgM).
ABOUT US
WHO WE ARE
Established in 1998, Anamol Laboratories Pvt. Ltd. today is synonymous with Quality and Consistency. The very name of Anamol comprises two separate words combined together as one which are “ANA” and “MOL” which means “ANALYSIS of MOLECULES”, explaining our core strength in understanding Chemistry and Biochemistry as a subject and how different molecules interact with each other. It is because of this in-depth knowledge that we can elevate our products to international standards or better. The power of current knowledge base and zeal to acquire new knowledge with the same depth and clarity has helped shape our core competency and gain trust from people within and outside the industry for close to two decades now.
WHAT WE DO?
We are highly sophisticated and professional manufacturers of Clinical Chemistry Reagents, Rapid Immunoassays, Urine Tests Strips, Immunology reagents, Dyes and Stains for Fluorometry and many others. Every product we develop, we keep in mind how we would help spread the confidence of an accurate diagnostic report to the Medical Practitioner as well as to the Patient. It is because of this goal at our hearts, we believe in adding value to the product to remain competitive and improve our Value Proposition.
Core Team
CHAIRMAN AND MANAGING DIRECTOR
Mr. Mahendra Dashora holds Master of Science in Chemistry (1979) and Master of Science in Biochemistry (1981) from the prestigious Maharaja Sayajirao University of Baroda (M.S.U), Vadodara, Gujarat (India). He has over 36 years of vast experience in the field of In Vitro Diagnostic (IVD) industry. He is the promotor of Anamol Laboratories Pvt. Ltd. and is a true visionary. He has 3 years (1981-1984) of exposure at Bayer Diagnostics (now Siemens Ltd.) as Chief Scientific Officer and In-charge of Quality Assurance.
He co-founded Accurex Biomedical Pvt. Ltd. in 1984 and took the responsibility of Director – Technical for 20 years. He was solely responsible for the development and manufacturing of all the products at Accurex. After parting with Accurex in 2004, he took over 100% shares of Anamol Laboratories Pvt. Ltd. and started the manufacturing facility at the present location.
At Anamol, his manufacturing capabilities and standardization grew in the fields of Immunology as well (Rapid Immunoassays, ELISA, CLIA, etc.)
Some of his achievements include:
1. First time indigenously manufactured enzyme based lyophilized Clinical Chemistry kits in India (1984).
2. First time indigenously manufactured liquid stable Clinical Chemistry reagents in India (1992).
3. First time indigenously manufactured Urine Test Strips in India (1984).
His passion to learn and zeal to make the most complex technologies available for the common mass brings a lot of wisdom in the organization.



We Recommend
New Arrivals
New products from manufacturers at wholesale prices