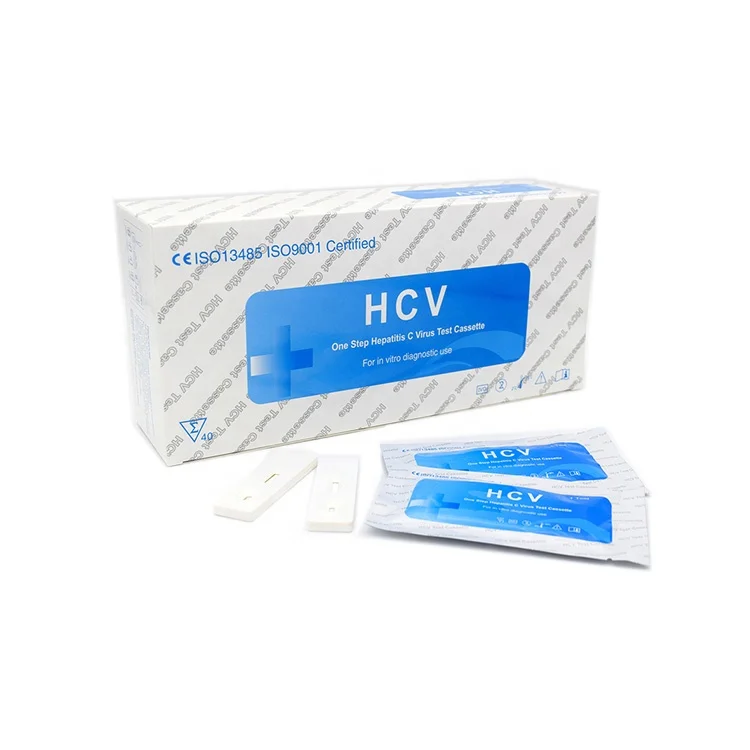
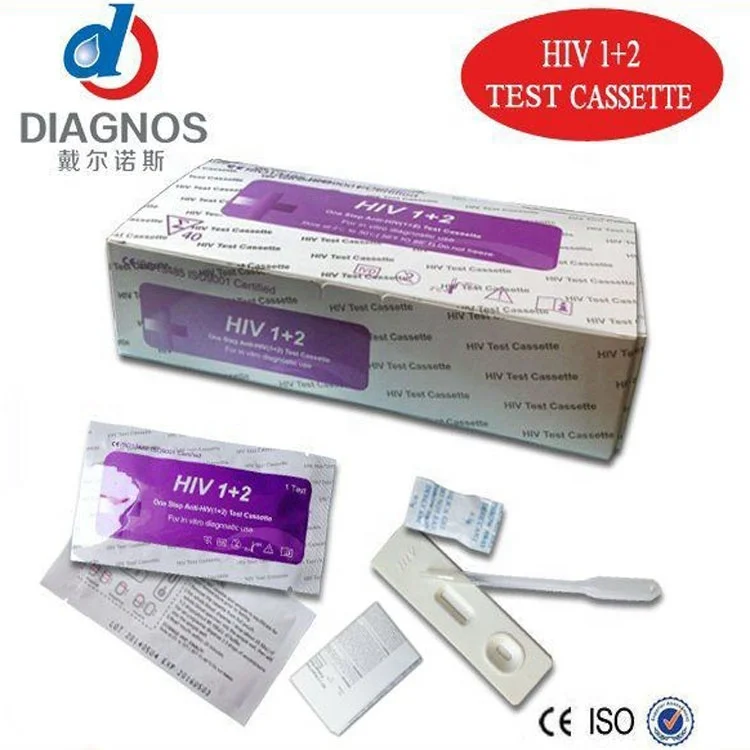
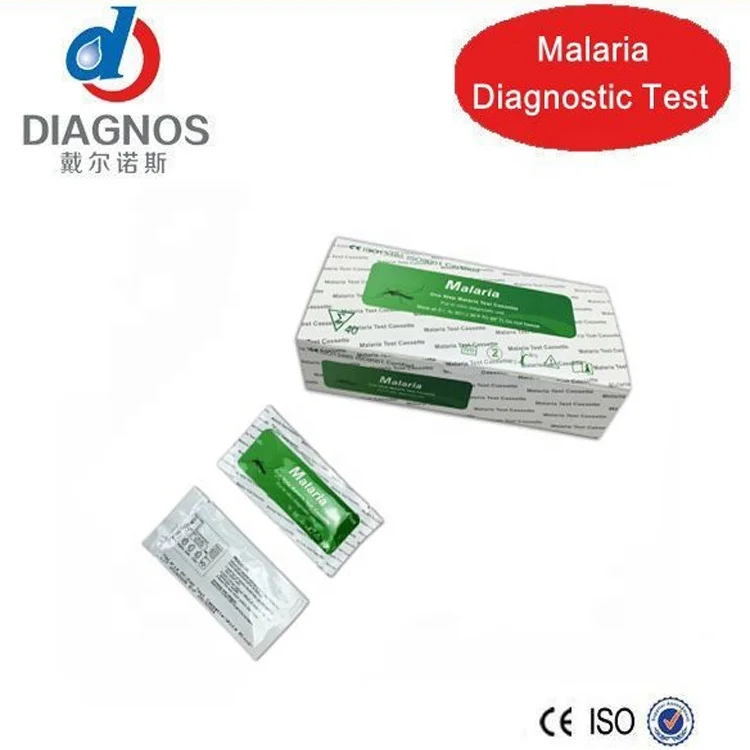
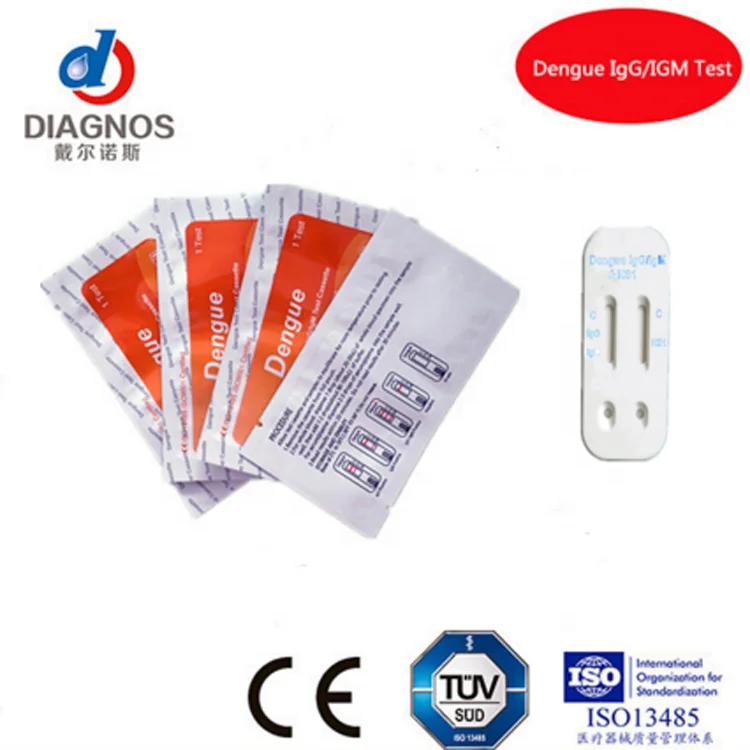
One Step Medical diagnostic Rapid Test HIV/HCV/syphilis/Malaria/Dengue
- Category: >>>
- Supplier: Nantong Diagnos Biotechnology Co. Ltd. [Jiangsu China]Nantong
Share on (62154403748):
Product Overview
Description
One Step Medical diagnostic Rapid Test HIV/HCV/syphilis/Malaria/Dengue
|
Product name |
One Step Medical diagnostic Rapid Test HIV/HCV/syphilis/Malaria/Dengue |
|
Shelf time |
24-36 month |
|
OEM time |
15-35 days |
|
Certificate |
CE/ISO13485/FSC |
|
Specimen |
whole blood |
|
Format |
Cassette |
|
Size |
Strip: 2.5mm/3.0mm |
|
|
Cassette :3.0mm/4.0mm |
|
Usage |
Read results within 10-15 minutes |
|
INTRODUCTION
For whole blood: Collect whole blood into a collection tube by venipuncture. If specimens are not immediately tested, they should be refrigerated at 2-80C. Freezing is recommended if the storage periods are more than 3 days. They should be brought to room temperature before testing. Using the specimen after long-term storage of more than three days can cause non-specific reaction. When stored at 2-8 0C, the whole blood sample should be used within three days.
ASSAY PROCEDURE Step 1: Bring the specimen and test components to room temperature if refrigerated or frozen. Mix the specimen well prior to assay once thawed. Step 2: When ready to test, open the pouch at the notch and remove device. Place the test device on a clean, flat surface. Step 3: For whole blood sample: Fill the dropper with the specimen then add 1 dropper of specimen(about 10uL). Then add 2 drops (about 60-100 μL) of Sample Diluent immediately into the sample well (S well) Step 4: Read the result within 15 minutes. Don’t read results after 30 minutes. Negative Only the C band shows color development. The T band shows no color development. Positive Both C and T bands show color development. The appearance of any burgundy color in the T band, regardless of intensity, must be considered as presence of the band. Invalid: If no C band is developed, the assay is invalid regardless of color development on the T band as indicated below. Repeat the assay with a new device. PRECAUTION 1. For in vitro diagnostic use only. 2. Do not use test kit beyond expiry date. 3. The test device should not be reused.
|












Packing Details : Standard packing ,neutral packing, your requirement are available
Delivery Details : 7-25 days after receiving the 30% deposit
Shipping Details : DOOR TO DOOR, BY AIR, BY SEA etc.
Payment Details :T/T,Western Union and Paypal are available.
After service:
1.Service tenet: quick and decisive and thoughtful thorough and accurate;
2.Service objectives: service quality to win customer satisfaction;
3.Service efficiency: We will have a dedicated staff to detect problems with the product for testingf if you are not satisfied when you receive your goods, and promptly get back to you within 24 hours.
4.Service principles: products in the course of product issues caused by improper operation, we will provide a solution.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices