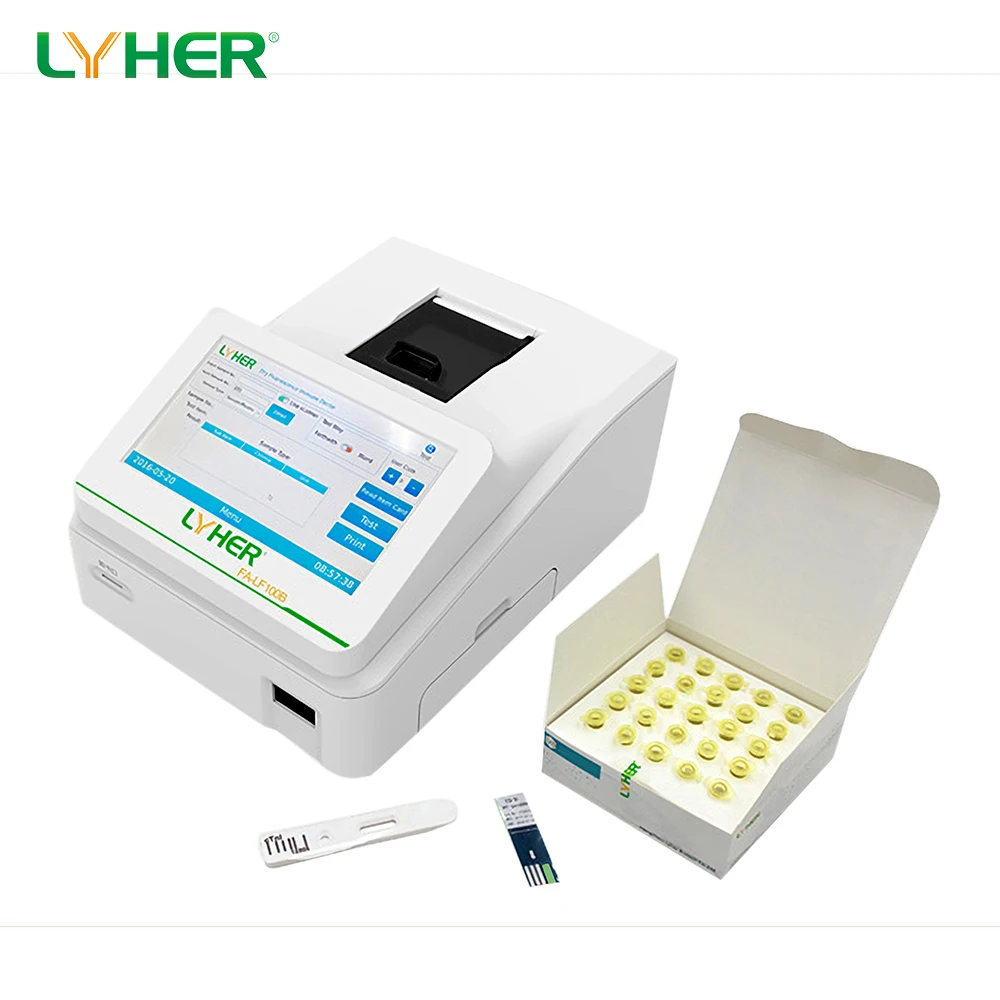
Cardiac Marker Troponin i Quantitative diagnose CTnI rapid test kit
- Category: >>>
- Supplier: Hangzhou Laihe Biotech Co. Ltd.
Share on (62250101188):
Product Overview
Description


Product Information
Product Name | Diagnostic Kit for the Quantitative Determination of Cardiac troponin I (Flourescent lateral flow immunoassay) |
Format | Cassette |
Test Item | Troponin I (cTNI) |
Specimen | Whole blood/Serum/Plasma |
Storage | 2-30°C |
Specification | 25 pcs/box |
Matched Machine |
Materials Provided
(1) Test card consists of paper strip and plastic shell. The main components on tha paper strip include: cellulose nitrate film, water absorbent paper, PS bottom plate; The membrane of cellulose nitrate was coated with rat anti-human Troponin I monoclonal antibody and sheep rabbit IgG.
Specification: 1pcs/box
B Box:
(3) Detection buffer: Fluorescent markers of Troponin I monoclonal antibodies, IgG Fluorescent markers of rabbits and PBS composition.
Specification: 25 tubes/box
Materials required but not provided: pipette and dropper for sample adding



(1) Preparation: open the dry fluorescence immunoanalyzer of Hangzhou Laihe Biotech Co.,Ltd.
(2) Prior to testing reagent, buffer, reaction tube sealed refrigeration, ID card and sample return to room temperature under test (15-30℃),the recommended reagents is restored to room temperature and opended.
(3) Calibration: Confirm that the ID card is matched with the batch number of the reagent, insert ID card after correct, and click read ID card after entering the test interface, and the reagent can be detected after the calibration is completed.
(4) Add the sample:
① 75μl mixture into the reagent plate.
②Tube detection buffer injection method:If the samples were to be tested, the serum/plasma would be removed 75μl, if the whole blood sample is removed from 150μl to the detection buffer,fully mix (30s-1min), and absorb 75μl mixture into the reagent plate.
(5) Modle:
According to the type of sample, the serum/plasma mode or whole blood mode is selected in the sample type option on the dry fluorescence immunoanalyzer.
(6) Test:
①Standard test: when the reagent card is added, the device will be inserted immediately, then click the “test button” the system will automatically countdown, and the automatic reading card will give the test results.
②Instant test: After the reagent card is added, the external reaction of the machine is 12 minutes, after the reaction, the reagent card is inserted into the instrument. Click the “test button”, the system will automatically read the card and give the test results.
(7) Click “print” and the system will automatically print the test results on the printer paper.
(8) After test of the reagent card, the excess premix, the used tip and the excess clinical sample were inactivated.
Interpretation of Results
According to a sample of 200 healthy people, the Troponin I <0.3ng/mL
Note: It is suggested that the laboratory should determine the applicability of reference range according to the population, age, sex, and diet of different regions, and establish practical reference scope when necessary.
INTERPRETATION OF TEST RESULTS
cTnI ≥0.3 ng/mL, According to the patient’s clinical situation, confirm the patient again.




Company Instruction
Hangzhou Laihe Biotech Co., Ltd. (hereafter referred to as "Laihe Biotech") is a life science company specialized in R & D, manufacturing, marketing and service in point-of-care testing, monitoring and health technology industry, is committed to provide fast, accurate and reliable health detection products and services. Founded by Dr returned from United States in February 2012, today Laihe Biotech has a team of experienced scientists researching and developing diagnostic products.
At present, we have a wide range of products including Drug of abuse test kit, Cardiac marker test kit, Tumor marker test kit, Fertility test kit, Infectious diseases test kit, Torch test kit, POCT test kit, POCT Quantitative Immunofluorescence Analyzer, Hair Drug Trace Rapid Analyzer.
History
Hangzhou Laihe Biotech Co., Ltd. (hereafter referred to as "Laihe Biotech") is a life science company specialized in R & D, manufacturing, marketing and service in point-of-care testing, monitoring and health technology industry, is committed to provide fast, accurate and reliable health detection products and services.
Professional Team
Founded by Dr returned from United States in February 2012, today Laihe Biotech have a team of experienced scientists researching and developing diagnostic products, and have already built an independent POCT platform which including various rapid test products like Cardiac Marker Series (cTnI, NT-proBNP, H-FABP, etc.), Inflammation Series (ds-CRP, PCT, etc.), Tumor Marker Series (AFP, CEA, etc.), Diabetes Monitoring Series (HbA1c), Fertility Series (TORCH) and Infectious Diseases Series. We continue to be innovative, and promote in-vitro diagnostic products in a miniaturized, individualized, family-oriented and low-cost way.







Business license, production licence, ISO 13485, CE, FSC etc.

Warehouse and Shipment
We have warehouse in our factory.
1. International express: TNT, DHL, FedEx, UPS, EMS. (door to door)
2. Logistics company: transportation by land, by air and by sea (according to customer's request)
After-sale Service System
Marketing research& customers feedback help us to know customers' further need and to upgrade our products. Our R&D direction keep pace with the marketing need: OEM support include sensitive adjustment (DOA); customer made packing and so on. New product lines are always being developed.
Professional and patient technical support are our service standard, If you have any technical problems, please contact us by Hotline/ Email/Facebook , we will reply you within 12 hours (working days).
1. Are you manufacturer or trader ?
We're manufacturer and have our own factory, so our price is very competitive.
2. What product are you offering ?
With a wide range of tests, the eight most common tests are:
(1) Drug of abuse test kit
(2) Cardiac marker test kit
(3) Tumor marker test kit
(4) Fertility test kit
(5) Infectious diseases test kit
(6) TORCH test kit
(7) POCT test kit
(8) POCT Quantitative Immunofluorescence Analyzer
(9) POCT immunofluorescence analyzer
3. Can you offer samples and make customized products ?
Of course. For samples, lease pick up the models you like and inform us to do a PI for sample freight; we can customize the products as customer's request, such as cut-off (DOA), strip size, package, etc.
4. Where is your market ?
We got ISO 13485:9001 and CE certificate. Our main sales market is Asia, Europe, Africa, Middle East and Oceania.
5. What's the lead time after we confirm the order ?
Well, they depends on the quantity and package design of your order. Neutral packages are usually very fast, around 10-15 working days, while OEM packages usually takes 25-40 working days. Orders above 1,000,000pcs will need 2 months for first order.

We Recommend
New Arrivals
New products from manufacturers at wholesale prices