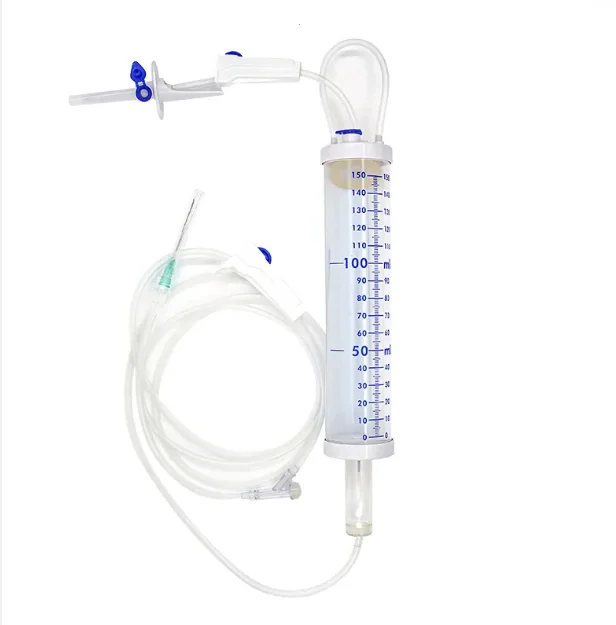
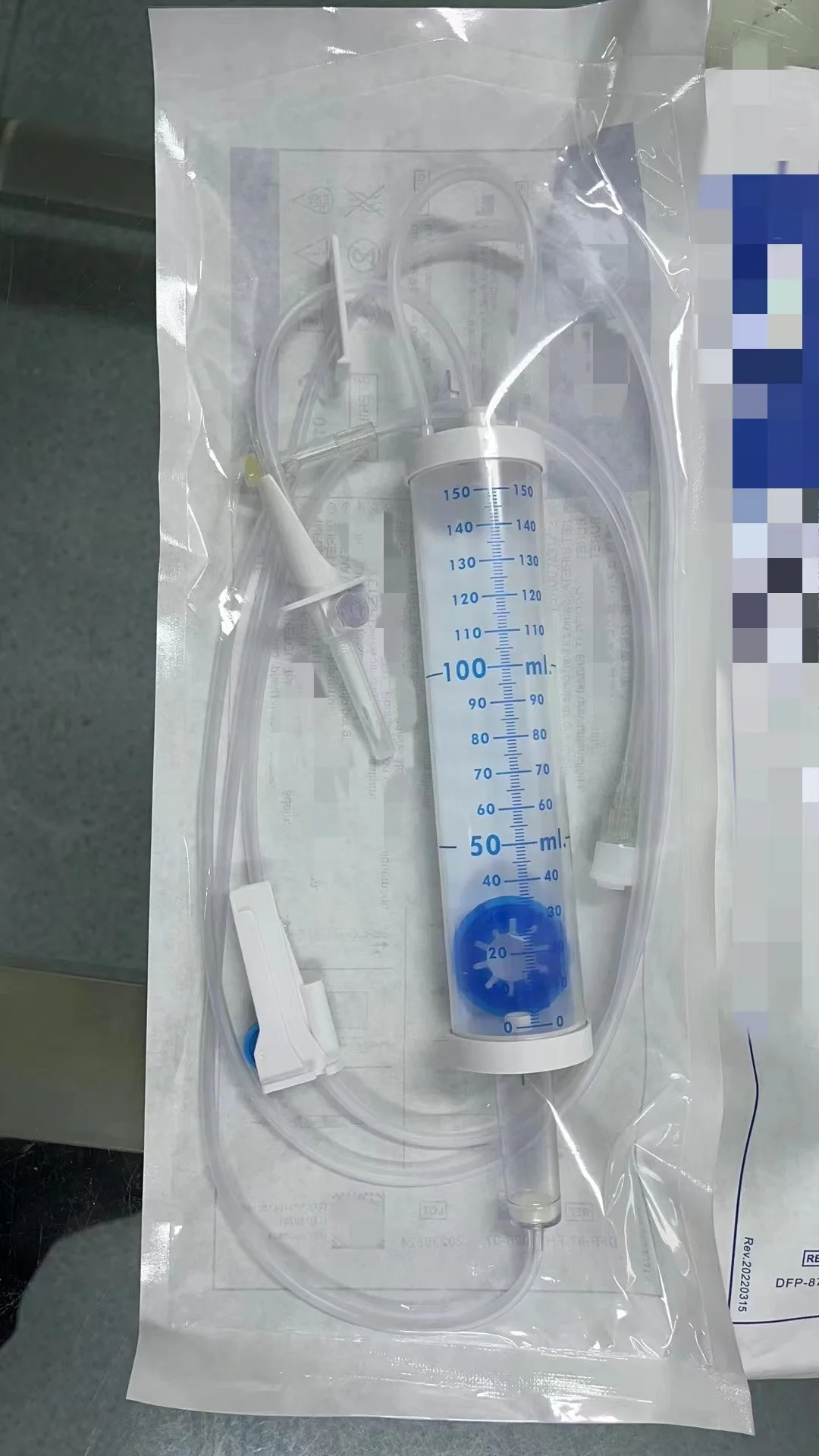
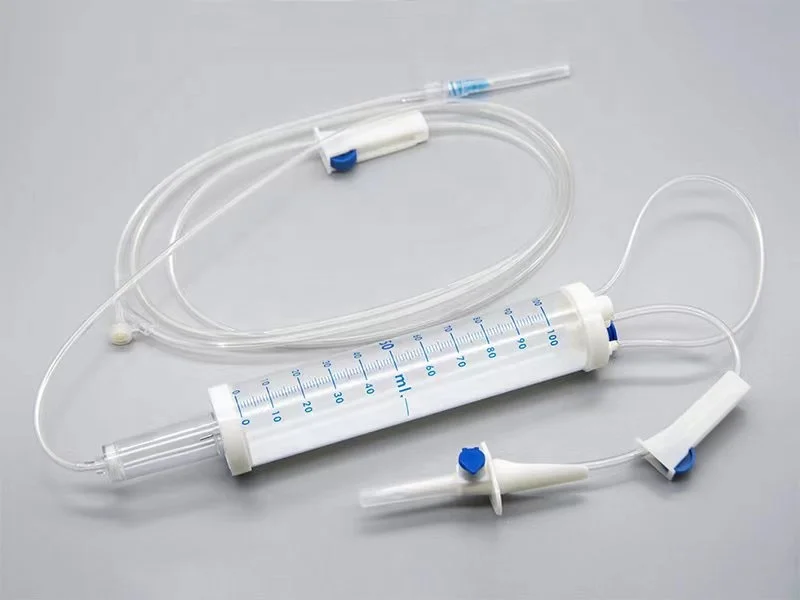
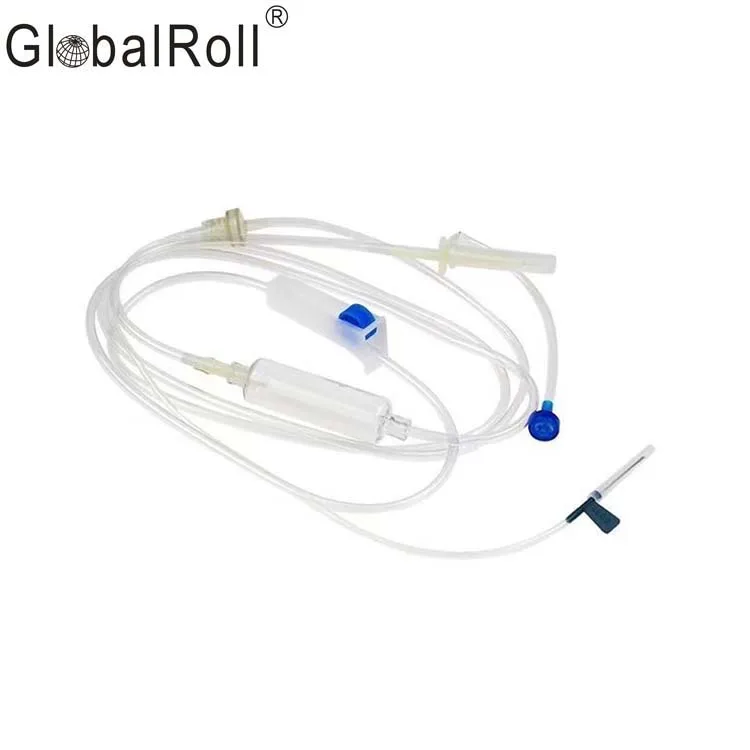
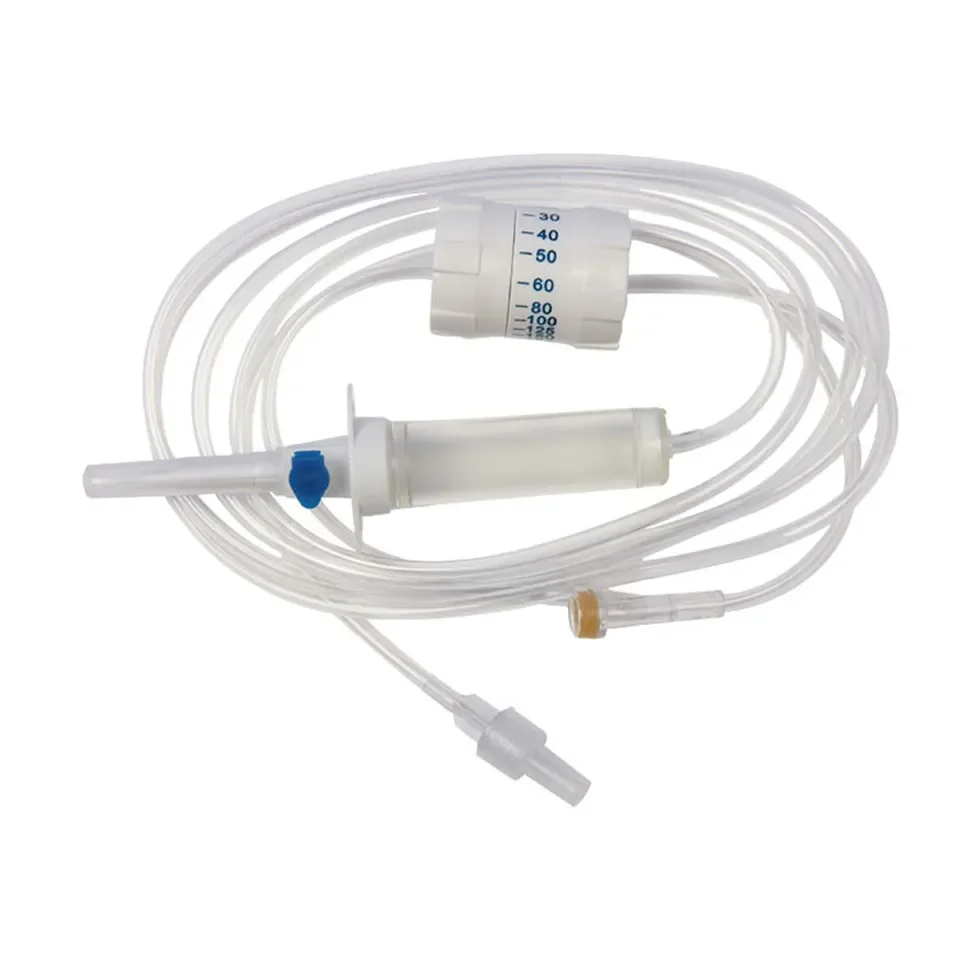
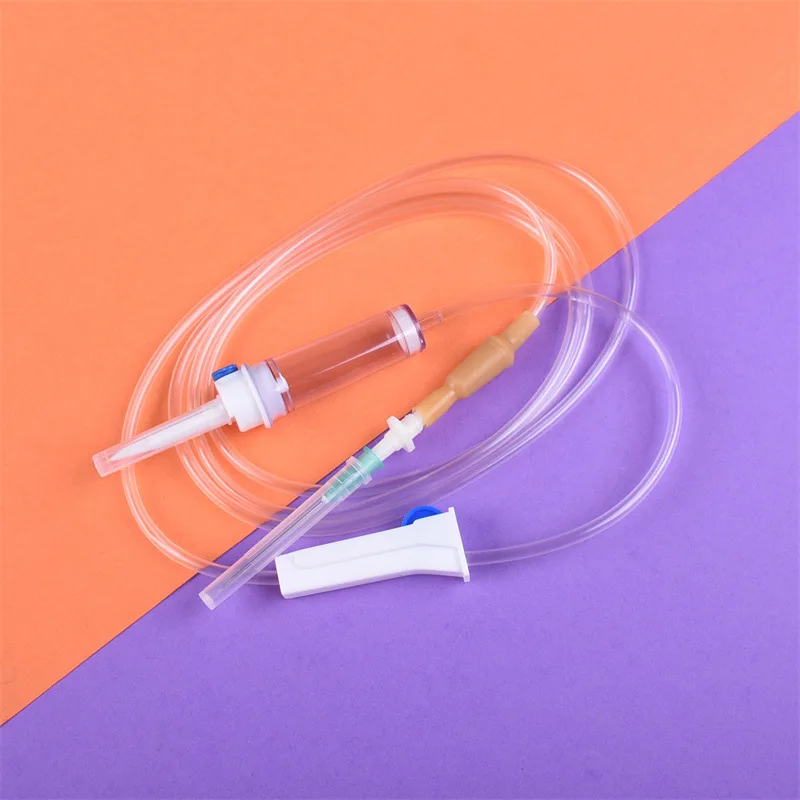
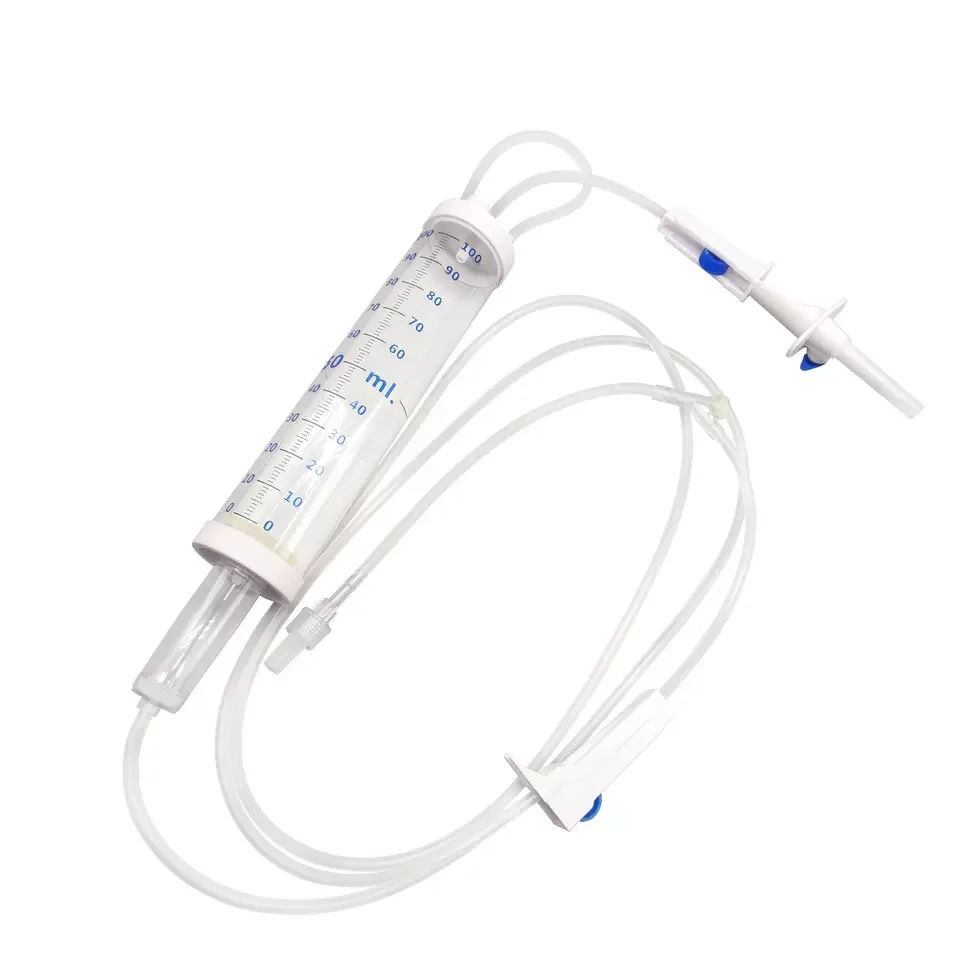
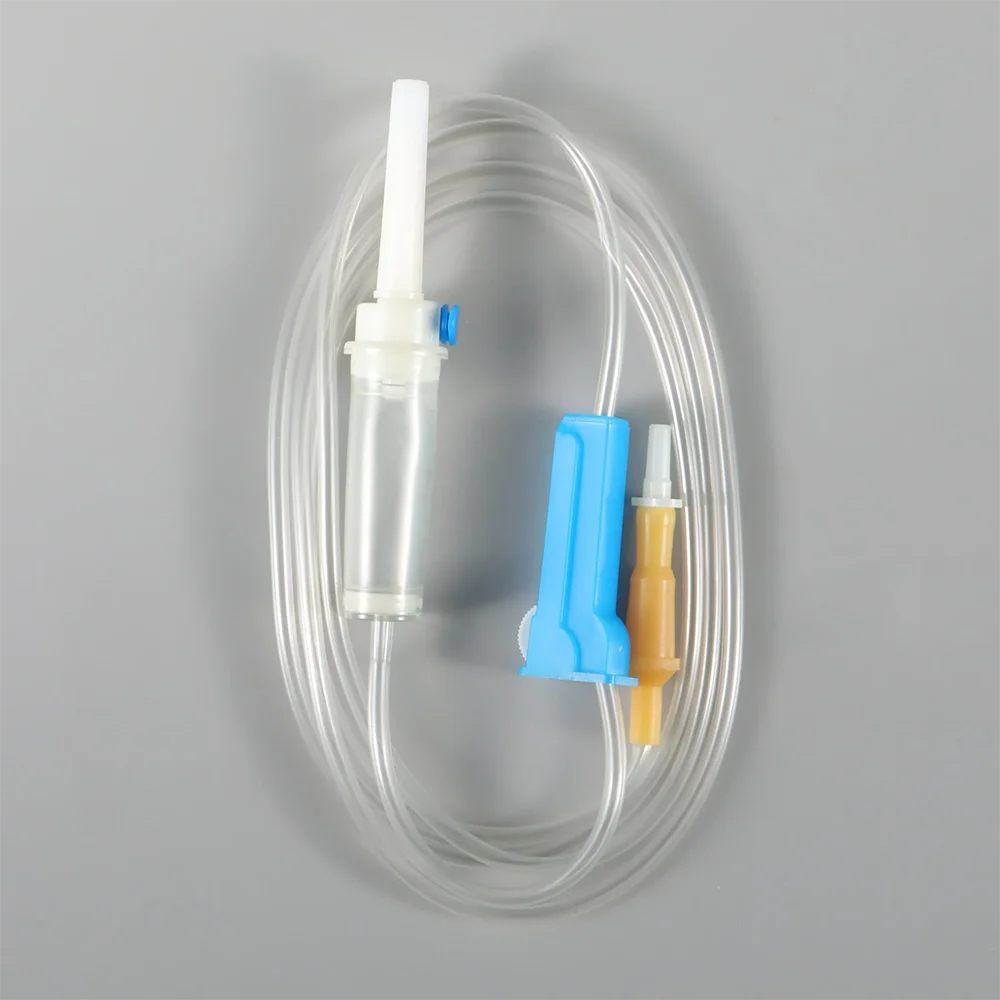
CE ISO13485 100ml disposable IV set with burette
- Category: >>>
- Supplier: Anhui Tiankang Medical Technology Co. Ltd.
Share on (62374539200):
Product Overview
Description
Product Description
Drip Chamber: PVC, sizes according to the ISO 8536-4 standards
Vented Spike: ABS
Flow Regulator: ABS or PP
Flexible Soft Tube: PVC, 1.5m Soft and kink resistant
Protective Cap: PP or PE, for closure piercing device with internal thread that prevents the bacteria from coming in
Air inlet: PE (with or without)
Fluid Filter: PVC (with or without)
"Y" Injection Port: PVC (with or without)
Injection site: Rubber (with or without)
Sharp Needle: Stainless steel (with or without), Hypodermic needle or Butterfly type needle available
Vented Spike: ABS
Flow Regulator: ABS or PP
Flexible Soft Tube: PVC, 1.5m Soft and kink resistant
Protective Cap: PP or PE, for closure piercing device with internal thread that prevents the bacteria from coming in
Air inlet: PE (with or without)
Fluid Filter: PVC (with or without)
"Y" Injection Port: PVC (with or without)
Injection site: Rubber (with or without)
Sharp Needle: Stainless steel (with or without), Hypodermic needle or Butterfly type needle available
Factory view




National government leaders visiting



Certifications

Exhibition

FAQ

Company Profile
Anhui Tiankang Medical Technology Co.,Ltd (Stock Abbreviation: Tiankang Medical, Stock Code: 835942)located in Tianchang City which is rated as the pearl of Anhui.It was founded as a subsidiary company of Anhui Tiankang Group in 1999. Our company have got official new three board on March 3, 2016 and now we have a registered capital of 57.78 million RMB yuan. We specialize in developing and producing products like safety syringes with retractable needles, auto disable syringes, I.V sets ,T.V sets ,extracorporeal blood circuits for haemodialysers,oral syringes, I.V catheter and intravenous infusion sets with burette etc. And now our company production more than 100 varieties of six series medical consumables products.
Our company has a factory of more than 600 acres holding a large scaled 100,000 class clean workshop 30,000 square meters.And we now have a staff of one thousand one hundred in including 430 technical engineers of middle and high ranges(about 39% of all the staff).Besides,we now have more than 100 first-class injecting machines and affiliated equipments of assembling and packing. We have two independent sterilization equipments and have established the internationally-advanced laboratory for biological and physical tests.
What deserves to be mentioned the most is that we are authorized European CE certificate (in 2002),ISO13485 certificate (in 2002) and WHO—PQS certificate (in 2010) which we hold as passports to enter international market. Our company's products began to be registered with the U.S. Food and Drug Administration in 2012 and currently rank among the best in the country for U.S. Food and Drug Administration-listed products. Autonomic products: safety syringes with retractable needles which registered by U.S. Food and Drug Administration 510 (k), disposable syringes and disposable hypodermic needles also passed U.S. Food and Drug Administration registered 510 (k). From June 6 ,2016 to June 9, with zero defect through the U.S. Food and Drug Administration field QSR 820 audit at the U.S. Food and Drug Administration site.
Our company has a factory of more than 600 acres holding a large scaled 100,000 class clean workshop 30,000 square meters.And we now have a staff of one thousand one hundred in including 430 technical engineers of middle and high ranges(about 39% of all the staff).Besides,we now have more than 100 first-class injecting machines and affiliated equipments of assembling and packing. We have two independent sterilization equipments and have established the internationally-advanced laboratory for biological and physical tests.
What deserves to be mentioned the most is that we are authorized European CE certificate (in 2002),ISO13485 certificate (in 2002) and WHO—PQS certificate (in 2010) which we hold as passports to enter international market. Our company's products began to be registered with the U.S. Food and Drug Administration in 2012 and currently rank among the best in the country for U.S. Food and Drug Administration-listed products. Autonomic products: safety syringes with retractable needles which registered by U.S. Food and Drug Administration 510 (k), disposable syringes and disposable hypodermic needles also passed U.S. Food and Drug Administration registered 510 (k). From June 6 ,2016 to June 9, with zero defect through the U.S. Food and Drug Administration field QSR 820 audit at the U.S. Food and Drug Administration site.
Related model



We Recommend
New Arrivals
New products from manufacturers at wholesale prices