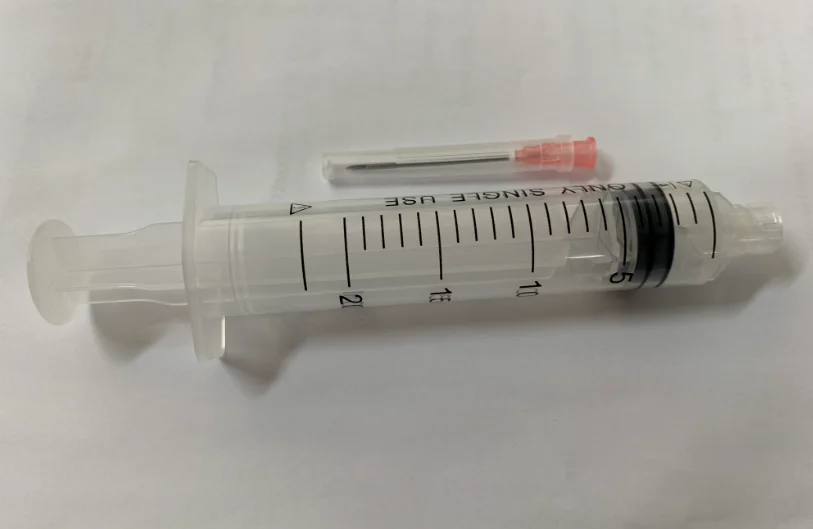
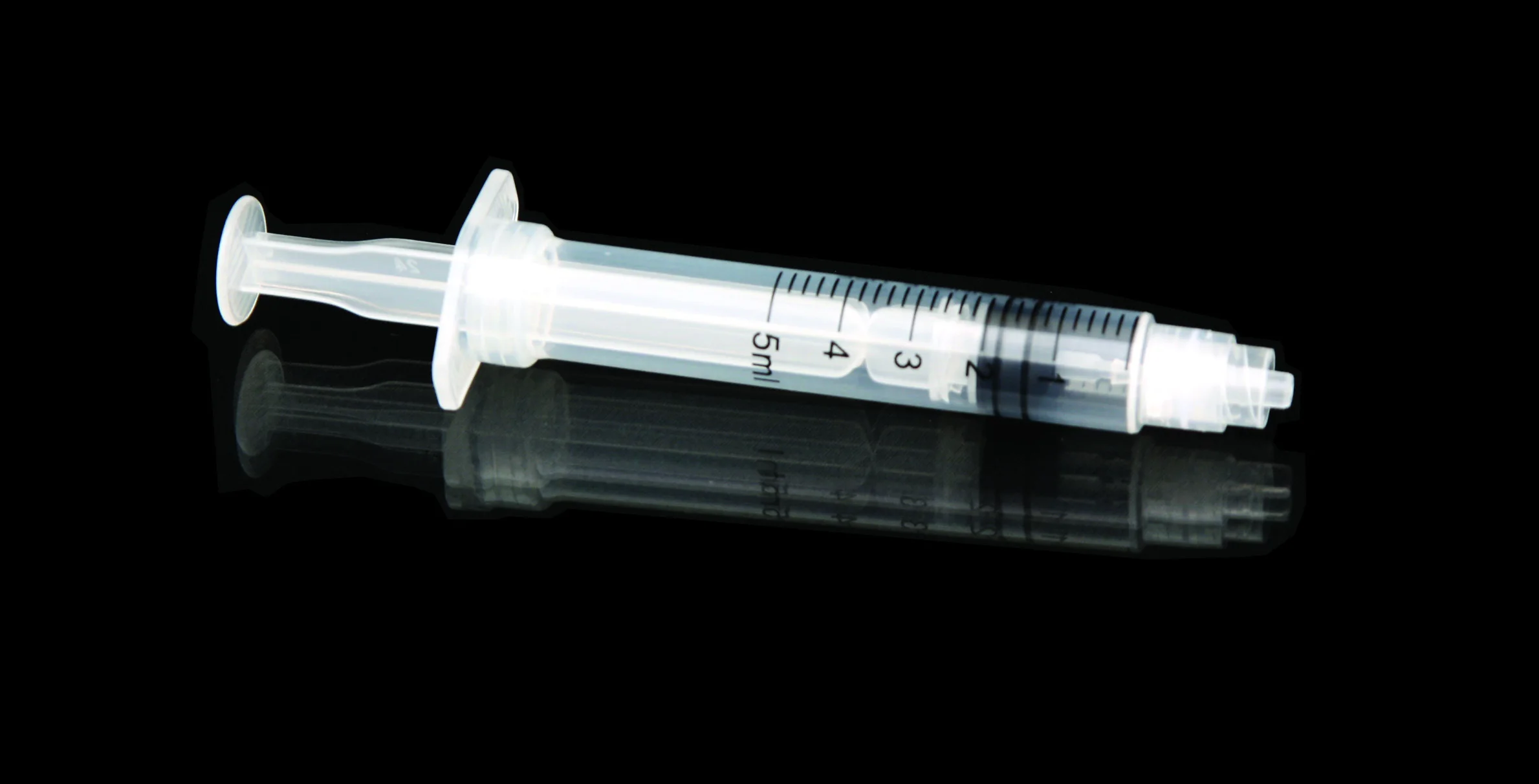
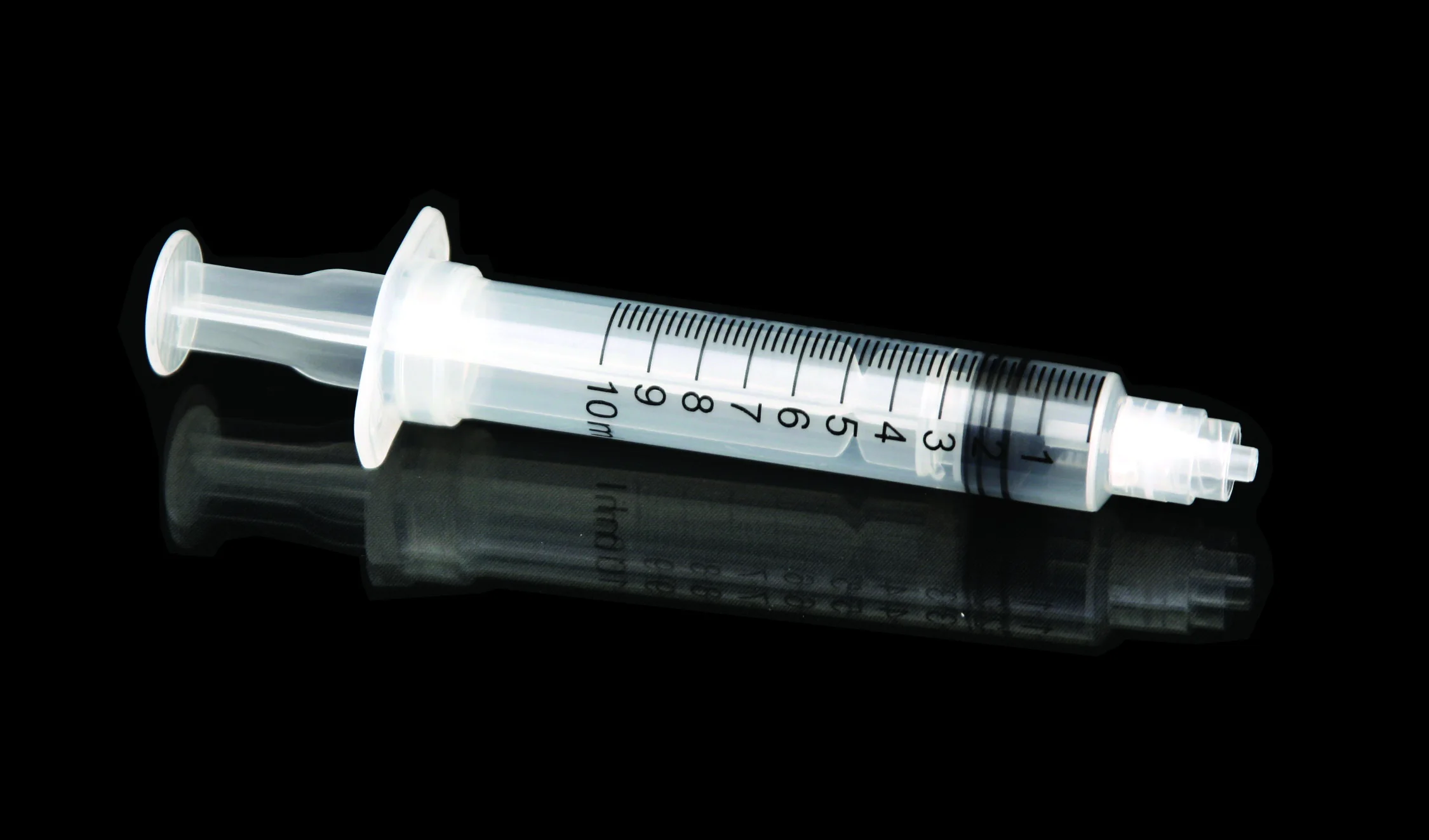
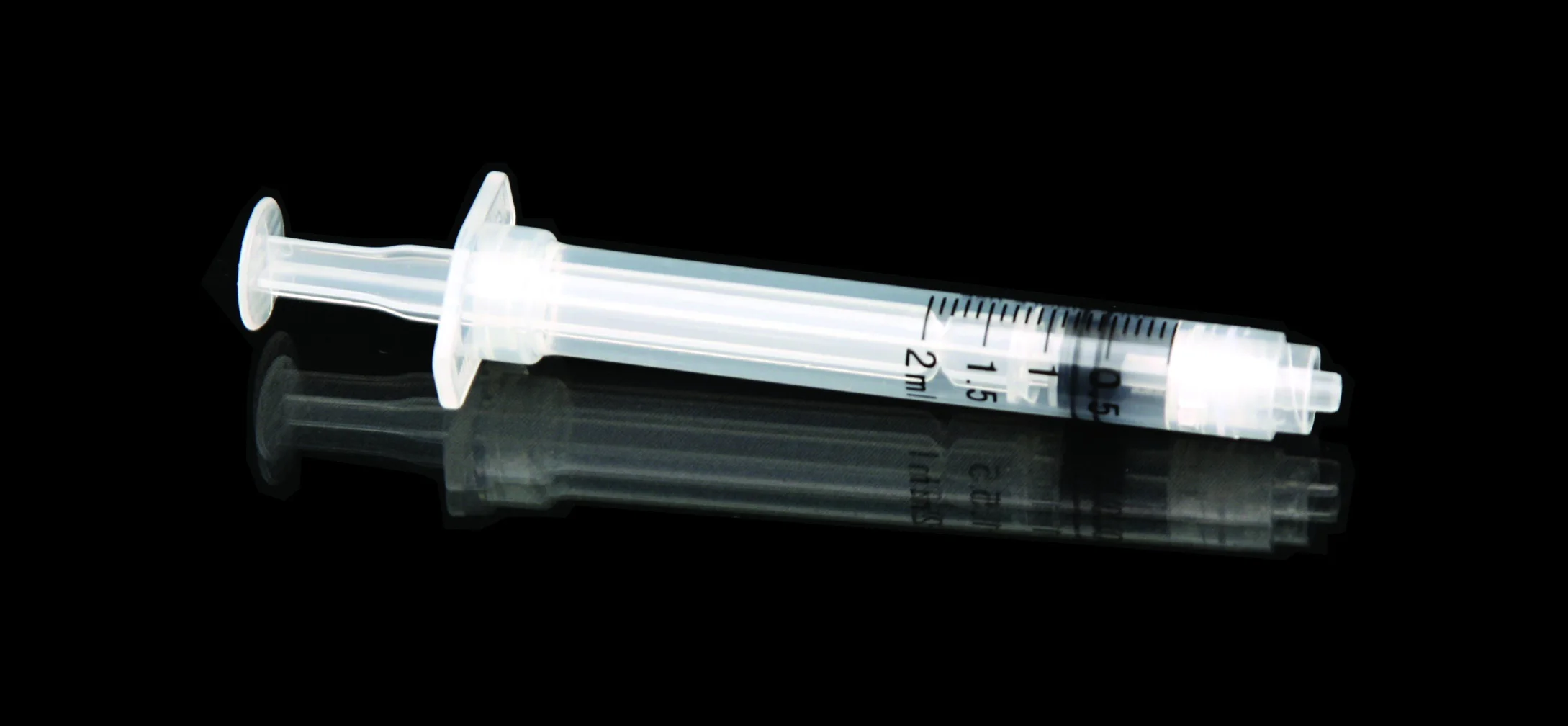
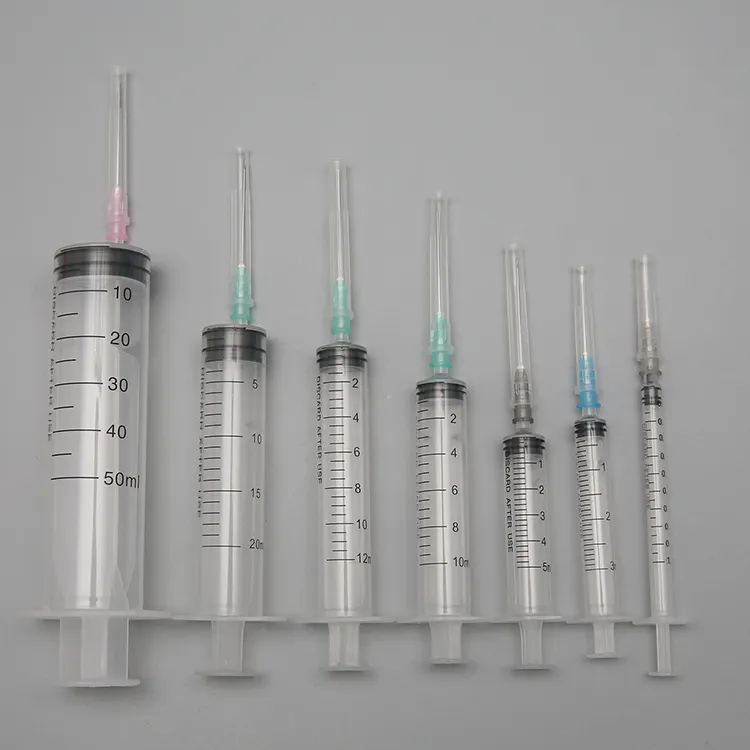
High quality 20ml safety syringe with luer lock
- Category: Disposable Syringe >>>
- Supplier: Anhui Tiankang Medical Technology Co. Ltd.
Share on (62384693560):
Product Overview
Description
Product Description

Product introduction:RPF as well as feature of manual operation will start spontaneously in order to prevent it from reusing.Meanwhile,it is able to prevent it from hurting people by covering hypodermic needle with barrel.
Why choose us?
1. We have the first and the most advanced glass syringe production line in Asia and have served more than 1000 customers all over the world, so we are quite professional and experienced.
2.OEM service, custom logo and packaging box are available.
3. 24-hour service all year round, feel free to contact us at any time.
2.OEM service, custom logo and packaging box are available.
3. 24-hour service all year round, feel free to contact us at any time.
Product packaging


National government leaders visiting



Exhibition


Our company has been working with NEOMED company and BD company for a long time. NEOMED and BD highly praise the quality and after-sales service of our infusion sets and syringes.
Company Profile

Anhui Tiankang Medical Technology Co.,Ltd (Stock Abbreviation: Tiankang Medical, Stock Code: 835942)located in Tianchang City which is rated as the pearl of Anhui.Our company have got official new three board on March 3, 2016 and now we have a registered capital of 57.78 million RMB yuan. We specialize in developing and producing products like safety syringes with retractable needles, auto disable syringes, I.V sets ,T.V sets ,extracorporeal blood circuits for haemodialysers,oral syringes, I.V catheter and intravenous infusion sets with burette etc.
Our company has a factory of more than 600 acres holding a large scaled 100,000 class clean workshop 30,000 square meters.And we now have a staff of one thousand one hundred in including 430 technical engineers of middle and high ranges(about 39% of all the staff). Besides, we now have more than 100 first-class injecting machines and affiliated equipments of assembling and packing. What deserves to be mentioned the most is that we are authorized European CE certificate (in 2002),ISO13485 certificate (in 2002) and WHO—PQS certificate (in 2010) which we hold as passports to enter international market. Our company's products began to be registered with the U.S. Food and Drug Administration in 2012 and currently rank among the best in the country for FDA-listed products. From June 6 ,2016 to June 9, with zero defect through the U.S. Food and Drug Administration field QSR 820 audit at the U.S. Food and Drug Administration site.
Our company has a factory of more than 600 acres holding a large scaled 100,000 class clean workshop 30,000 square meters.And we now have a staff of one thousand one hundred in including 430 technical engineers of middle and high ranges(about 39% of all the staff). Besides, we now have more than 100 first-class injecting machines and affiliated equipments of assembling and packing. What deserves to be mentioned the most is that we are authorized European CE certificate (in 2002),ISO13485 certificate (in 2002) and WHO—PQS certificate (in 2010) which we hold as passports to enter international market. Our company's products began to be registered with the U.S. Food and Drug Administration in 2012 and currently rank among the best in the country for FDA-listed products. From June 6 ,2016 to June 9, with zero defect through the U.S. Food and Drug Administration field QSR 820 audit at the U.S. Food and Drug Administration site.
Factory view




Shippment


Recommend Products


Auto disable syringe has been in a design that RPF will start all by itself just after it has been used in order to prevent it from reusing.Luer slip type is also used widely in medical work.
We Recommend
New Arrivals
New products from manufacturers at wholesale prices