Class II Instrument classification blood glucose test strips
- Category: >>>
- Supplier: Wuhan J.H. Bio-Tech Co. Ltd.
Share on (62407896775):
Product Overview
Description
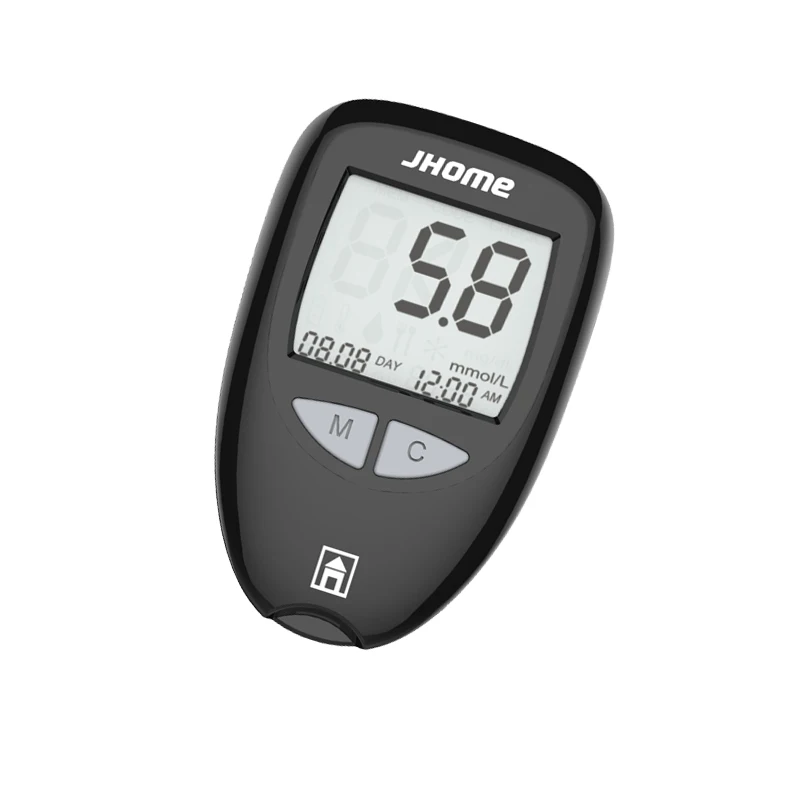
Class II Instrument classification blood glucose test strips

Application:This product is used for in vitro monitoring, the test results are only used to monitor the effect of blood glucose control in diabetic patients, can not be used as a diagnostic and screening tool for diabetes.
Principle:This product uses the bio sensor technology based on the principle of glucose oxidant micro-current measurement technology, by testing the blood glucose test strip in the glucose oxidant and glucose in the blood samples produced by the chemical reaction generated by the micro-current Size to determine and show the glucose concentration in the blood sample.
Specification:
Blood glucose meter feature | |
Model | JHBGM001A |
Silence | 20142402059 |
Blood sample type | fresh capillary whole blood |
Calibration | No Coding (i-code) |
Measuring range | 1.1-33.3mmol/L |
Measuring Unit | mmol/L(mg/dL) |
Battery life | 1000 testing |
Power supply | CR 2032*1 |
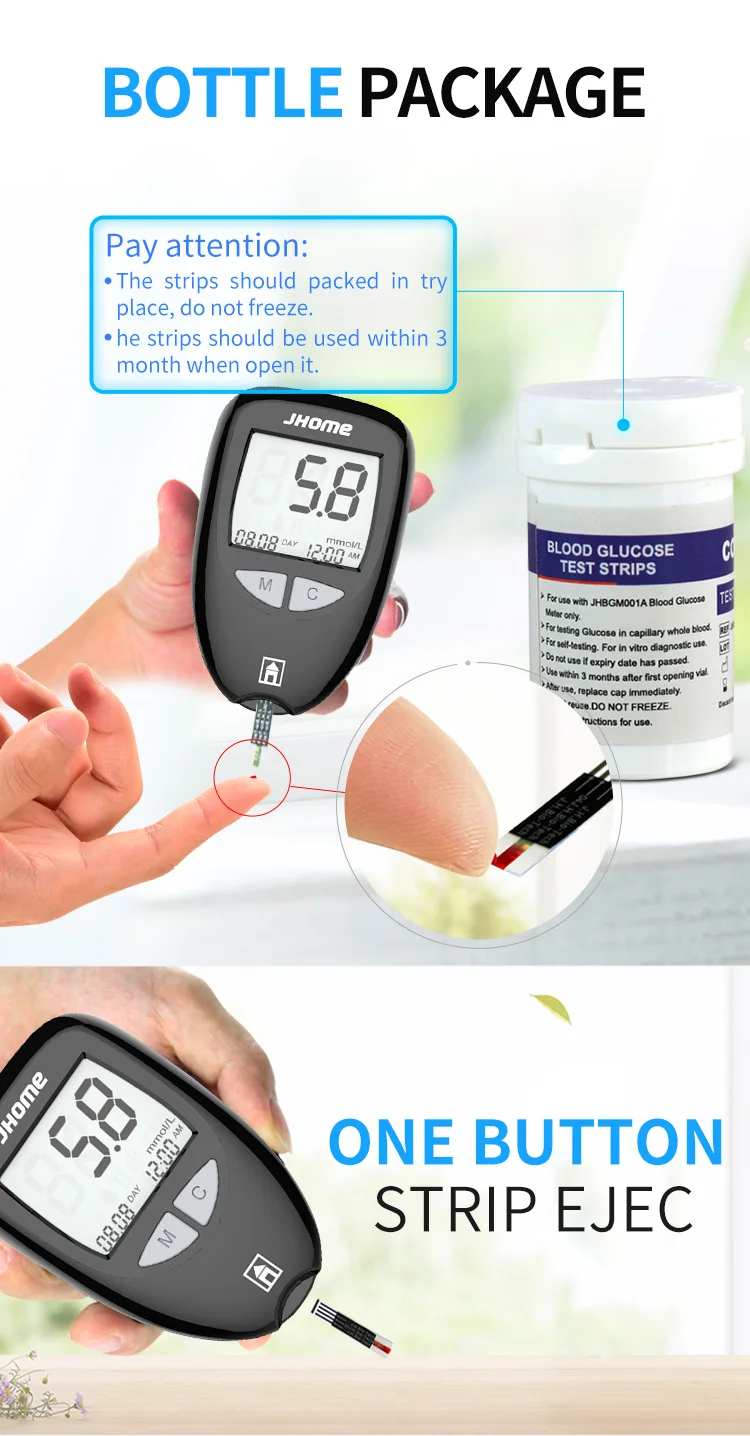
Sample volume | 0.5µL |
Reaction time | 5 seconds |
Date and time display | YES |
Alternate sit testing | YES |
PC connection | YES |
Automatic shutdown | after 100 seconds without key operation |
Measuring temperature | 10-40℃ |
Measuring humidity | 10%-90% |
Strip Valid | 24months |
guarantee period | 5 years |
Size | 15*54*86mm |
Weight | 45g (including battery) |



24hours services

Q1: Are you manufacturer?
A1: Yes, our factory has been for 10 years. Sincerely welcome to visit our factory any time.
Q2: The company established time and where are you?
A2: Our company was established in 2005, and specialized in Medically Approved Products, Connected Healthcare Devices more than10 years. Our company located in middle China--Wuhan City, Hubei provice.
Q3: Do you have any factory audits?
A3: Yes,now we have passed ISO13485, CE0123, ROHS etc.
Questions of Order:
Q1: Can you provide OEM or ODM service?
A1: Yes, we have strong developing team to support OEM and ODM service. The products can be made according to your request.
Q2: What's your MOQ?
A2: 500pcs. For our first cooperation, your small order is acceptable, but the price is a little highly, you know, the price is based on quantity.
Q3: What is the sample lead time & production lead time?
A3: the stock sample 1 day, making sample 5-7 workdays after received your sample charge.The production lead time depend on your order quantity based on MOQ.
Q4: What's your payment terms and trade terms?
A4: T/T 30% as deposit, and 70% before delivery. We'll show you the photos of the products and packages before you pay the balance.
Q5: What is your terms of delivery?
A5: FOB/CIF/By air as you like.






We Recommend
New Arrivals
New products from manufacturers at wholesale prices