High density USP/EP standard Glutathione powder , L Glutathione reduced
- Category: Vitamins >>>
- Supplier: Sinoway Industrial Co. Ltd. [Fujian China]Sinoway Ltd.
Share on (971419823):
Product Overview
Description
High density USP/EP standard Glutathione powder , L-Glutathione reduced
1. Basic information of L-Glutathione reduced :
1.Product name: L-Glutathione Reduced
2.CAS No.:70-18-8
3. Specification : 1) Food grade USP41 ; 2) pharmaceutical /injection grade EP7
4.MF:C20H32N6O12S2
5.MW:612.63
6. Purity :98%-101% by HPLC
7. Documents : GMP /DMF for pharamceutical /injection grade
2.Our advantages of L-Glutathione reduced :
1). Excellent quality: assay 98% up ,conform to USP41 standard ,
high density ( Bulk density :0.55g/ml ,Tapp density: 0.65g/ml).
2). Lowest price : Lowest price in the market ( different quantities with different prices) .
3). Large capacity & stock : 3-4 tons per month and usually with 500kg in stock for prompt shipment
4). Better payment terms : T/T 30days after shipment, accept Alibaba Trade Assurance
5) Various shipping ways : by air./ by sea / by courier (FedEx) , we have warehouse in USA.
3.Application of L-Glutathione reduced :
L-Glutathione Reduced is widely used in Pharmaceuticals, Nutraceuticals,Health functional foods, Dietary supplements, Skin-Whitening products, Cosmetic etc
The regular dosage form: Lyophilized Powder for Injection,Tablets,Capsules, Eye drops etc.
4. Functions of L-Glutathione reduced :
a.Daily nutraceuticals & Dietary supplement --(food /cosmetic grade)
1) Anti-aging, antioxidant, maintain skin vigor and luster.
2) Whitening skin: restraining the melanin
3) Improve immunity: enhance immune cells function effectively prevent viruses.
b.Drug treatment and prevention--(pharmaceutical grade)
1) Protect liver: prevention & treatment of liver diseases.
2) Detoxification: drugs and other kinds of poisoning auxiliary treatments,helpful for excreting toxins.
3) Prevention and treatment of eye diseases.
4) Auxiliary treatment of diabetes.
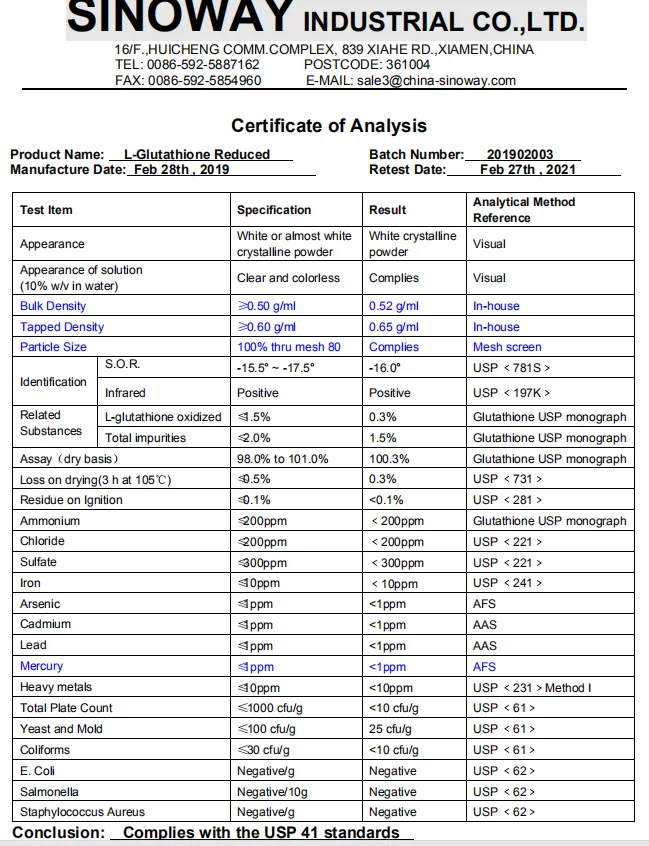
5. COA of L-Glutathione Reduced (USP41) :
|
Test Item |
Specification |
Result |
Analytical Method Reference |
|
|
Appearance |
White or almost white crystalline powder |
White crystalline powder |
Visual |
|
|
Appearance of solution (10% w/v in water) |
Clear and colorless |
Complies |
Visual |
|
|
Bulk Density |
≥0.50 g/ml |
0.52 g/ml |
In-house |
|
|
Tapped Density |
≥0.60 g/ml |
0.65 g/ml |
In-house |
|
|
Particle Size |
100% thru mesh 80 |
Complies |
Mesh screen |
|
|
Identification |
S.O.R. |
-15.5° ~ -17.5° |
-16.0° |
USP <781S> |
|
Infrared |
Positive |
Positive |
USP <197K> |
|
|
Related |
L-glutathione oxidized |
≤1.5% |
0.3% |
Glutathione USP monograph |
|
Total impurities |
≤2.0% |
1.5% |
Glutathione USP monograph |
|
|
Assay(dry basis) |
98.0% to 101.0% |
100.3% |
Glutathione USP monograph |
|
|
Loss on drying(3 h at 105℃) |
≤0.5% |
0.3% |
USP <731> |
|
|
Residue on Ignition |
≤0.1% |
<0.1% |
USP <281> |
|
|
Ammonium |
≤200ppm |
<200ppm |
Glutathione USP monograph |
|
|
Chloride |
≤200ppm |
<200ppm |
USP <221> |
|
|
Sulfate |
≤300ppm |
<300ppm |
USP <221> |
|
|
Iron |
≤10ppm |
<10ppm |
USP <241> |
|
|
Arsenic |
≤1ppm |
<1ppm |
AFS |
|
|
Cadmium |
≤1ppm |
<1ppm |
AAS |
|
|
Lead |
≤1ppm |
<1ppm |
AAS |
|
|
Mercury |
≤1ppm |
<1ppm |
AFS |
|
|
Heavy metals |
≤10ppm |
<10ppm |
USP <231>Method I |
|
|
Total Plate Count |
≤1000 cfu/g |
<10 cfu/g |
USP <61> |
|
|
Yeast and Mold |
≤100 cfu/g |
25 cfu/g |
USP <61> |
|
|
Coliforms |
≤30 cfu/g |
<10 cfu/g |
USP <61> |
|
|
E. Coli |
Negative/g |
Negative |
USP <62> |
|
|
Salmonella |
Negative/10g |
Negative |
USP <62> |
|
|
Staphylococcus Aureus |
Negative/g |
Negative |
USP <62> |
|

We Recommend
LY Health Supply Vitamin D3 10000 IU Vitamin D3 Powder
Factory Supplies High-quality Vitamin D3 Powder 100 000iu/g
Effervescent Vitamin C Serum Tablet 1000mg Ascorbic Acid
New Arrivals
New products from manufacturers at wholesale prices