Огромный спрос на лучшее качество одноразового использования iCARE Гепатит C (HCV) Набор для быстрого тестирования с 2 годами срока годности
- Категория: >>>
- Поставщик: JAL MEDICAL (SINGAPORE) PTE. LTD.
Сохранить в закладки 10000005043140:
Описание и отзывы
Характеристики

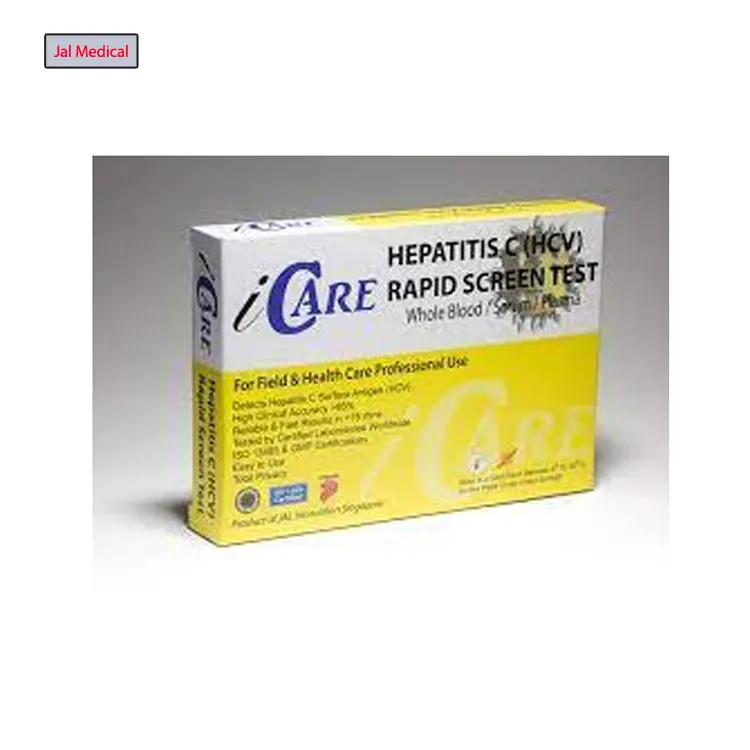
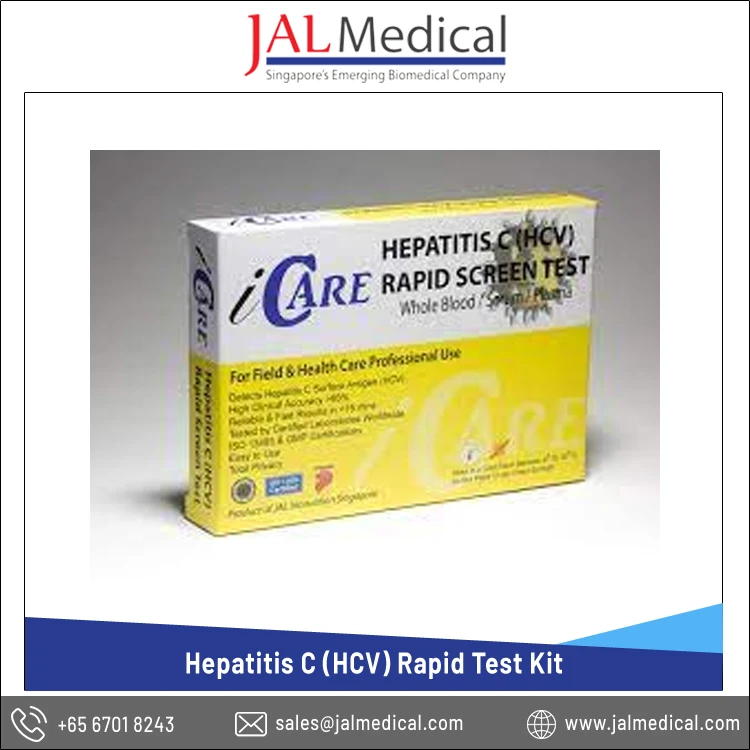
Huge Demand on Best Quality Single Time Use iCARE Hepatitis C (HCV) Rapid Test Kit with 2 Years Shelf Life
iCare Anti-HCV Rapid Test Kit is a screening test for the qualitative detection of the anti-HCV antibodies in whole blood as an aid in the diagnosis of Hepatitis C infection. Hepatitis C is a liver disease caused by the hepatitis C virus. The virus can cause both acute and chronic hepatitis, ranging in severity from a mild illness lasting a few weeks to a serious, lifelong illness. It is most commonly transmitted through: injecting drug use through the sharing of injection equipment; the reuse or inadequate sterilization of medical equipment, especially syringes and needles in healthcare settings; and the transfusion of unscreened blood and blood products.
Precautions
1. Do not use the test kit beyond the expiration date.
2. Do not use the kit if the pouch is punctured or not well sealed.
3. For in vitro use only. Do not swallow.
4. All specimens from the body should be treated as potentially infectious.
5. Contaminated blood may give incorrect test results.
6. Discard after first use. The test cannot be used more than once.
DISPOSAL: The used-device has the risk of infection. Please dispose all used
contents properly.
Material Required But Not Provided: Timer
Storage And Stability: The kit must be stored at 2-30OC.
Performance Characteristics
1. Specificity:
The specificity of iCare Anti-HCV Test is based on clinical studies using confirmed negative serum samples from blood bank and hospital patients in USA (66 samples) and China (90 samples). The studies were performed comparing the results from Rapid Anti-HCV test and that from Abbott's ELISA as a reference test. The overall specificity was found to be 97 - 99%.
2. Sensitivity:
In the same studies mentioned above, iCare Anti-HCV Test was evaluated with 61 confirmed positive serum samples (USA: 31 samples and China: 30 samples). All 61 samples were found reactive.
Limitations:
This Test Is A Screening Test And All Positives Must Be Confirmed Using An Alternate Test Such As Western Blot. The Test Is Intended For Field And Professional Use Only.





Each Kit Contains:
- Test cards individually foil pouched with a desiccant
- Plastic dropper
- Sample diluent
- Safety lancet
- Alcohol swab
- Package insert


Product Name | Hepatitis C (HCV) Rapid Test Kit |
Packing | Hospital Version: 40 Tests Per Kit Field Version: 1 Per Foil |
Specimen | Whole blood / Serum / Plasma |
Type of Test | Antibody |
Format | Hospital version: 1 well cassette, pipette, diluents (2 bottles) Field version: Alcohol swab, lancet, 1 well cassette, pipette, diluents |
Sample | 10ul serum |
Result | Within 15 minutes |
Specificity | 100% |
Sensitivity | 100% |
Shelf Life | 24 months |
Storage | Store at 4 ºC ~ 30 ºC in the sealed pouch up to the expiration date. |

JAL Medical Singapore is a company of a public listed manufacturer OK Biotech Co. Ltd. Our head office is located in Hsinchu, Taiwan. We are ISO 13485 and GMP certified. Our ultimate goal is to serve our global partners with a truly integrated biomedical manufacturing facility with the offering of a wide range of proprietary diabetic monitoring systems and in-vitro diagnostic test kits.
The JAL Medical Group of companies started its medical devices business for more than 20 years and is now reputed to one of the world’s leading manufacturer and global distributor of rapid diagnostic test kits. The group is also one of the world’s global dominant manufacturer of blood glucose monitoring systems.
We are committed to serve our partners by manufacturing and distributing high quality, reliable and competitive products and services to every continent. Our vision is to provide safe, reliable and cost-effective solutions in markets around the world.
Our Group’s Key Business Divisions are:
- Manufacturing of an extensive range of Rapid Diagnostic Test Kits particularly a complete range of professional use and home use STD test kits.
- Manufacturing of an extensive range of professional and home POC use blood glucose monitoring systems.
- Provision of Professional Consultancy Services for the development of Rapid Diagnostic Test Kits Design and Production Facilities.
- Health Products – The group has now entered into a joint collaboration with Nutrisyz Singapore to develop and distribute globally an exciting range of Proprietary Medicine and Herb-Based Health Supplements. These high-efficacy Chinese Proprietary Medical (CPM) are approved and listed with the Singapore Health Science Authority (HSA).
LATEST NEWS
- We are pleased to announce that JAL Medical Group of Companies (JAL Medical Singapore & JAL Innovation Singapore) in close tandem with our South Africa partner Armada Diagnostics Pty Ltd have successfully won the South African National Dept of Health continually for 9 years in a row from 2014 to 2023.
- We are supplying our co-branded WHO pre-qualified HIV 1&2 diagnostic test kits. It is through a unified collaborative effort.
- We are exceptionally pleased to announce our compelling success in winning the 3rd consecutive tenders.
- These tenders were reputed to be the largest supply of diagnostic test kits tender in the world. During these years, more than 65 million diagnostic test kits were delivered and used successfully. During the years 2021 to 2023 new tender allotment, we are expected to deliver another 42 million diagnostic test kits.
- In addition, JAL Medical Singapore commands a good market share for our iCare Advanced blood glucose monitoring systems in Laos, Ivory Coast, Nigeria, Indonesia to name a few.
- Whereas our home use private testing STD test kits are supplied to key projects in Myanmar, Laos, New Zealand, Equatorial New Guinea, Thailand as well as in several Pacific and Caribbean Islands.
- Our strong teamwork, our global brand, consistent product quality and good support services have contributed to our global success.
- We take this opportunity to thank all our global partners for contributing to our growth for the past 20 years.
Check Out Our Tutorial Videos on How to Use Our Test Kits









Choose Your Language
Похожие товары
Пищевой сорт, Лучшая цена, D-глюкурон в порошке 32449-92-6, глюкуронолактон D-глюкуронолактон
TPA3116 D2 50 Вт + 50 Вт двухканальный цифровой стерео усилитель доска DC 24V XH-M189
Качество и надежность, одноразовое использование, ВИЧ 1 и 2, набор для быстрого тестирования полости рта из Сингапура
Наборы для теста на овуляцию LH, тест на беременность, онлайн-тесты
Высокоточный простой домашний набор для быстрого тестирования Chlamydia от надежного поставщика
Лето 2021, лидер продаж, машины для производства колы/машина для смешивания колы
Новое поступление защитный чехол силиконовый держатель Airtag ошейник для собаки кошелек брелок трекер чехол для Apple Airtags
Новые поступления
Новинки товаров от производителей по оптовым ценам